|
|
| | dilithium acetylide Basic information |
| Product Name: | dilithium acetylide | | Synonyms: | dilithium acetylide;lithium carbide;1,2-Dilithioacetylene;Dilithioacetylene;Ethyne-1,2-diylbislithium;Ethynylenebislithium | | CAS: | 1070-75-3 | | MF: | C2Li2 | | MW: | 37.9034 | | EINECS: | 213-980-1 | | Product Categories: | | | Mol File: | 1070-75-3.mol | 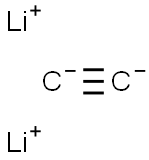 |
| | dilithium acetylide Chemical Properties |
| density | 1.650 | | solubility | reacts with H2O; insoluble in organic solvents | | color | white hygroscopic crystals, crystalline |
| | dilithium acetylide Usage And Synthesis |
| Chemical Properties | crystal(s) white powder(s); decomposes in water, evolves acetylene when dissolved in acid [HAW93] | | Preparation | Lithium carbide, Li2C2, may be prepared by the direct combination of lithium and
carbon at about 1000°C or by the reaction of lithium metal with acetylene in liquid ammonia.
The latter process first yields lithium acetylide, LiC=CH, which decomposes to lithium
carbide. The conversion to the carbide is incomplete, however . A convenient laboratory
preparation of lithium carbide may be carried out by bubbling acetylene into a solution of
n-butyllithium in hexane. The precipitated carbide may be filtered from the reaction
mixture, washed, and dried. Lithium carbide is ionic and yields acetylene on hydrolysis. |
| | dilithium acetylide Preparation Products And Raw materials |
|