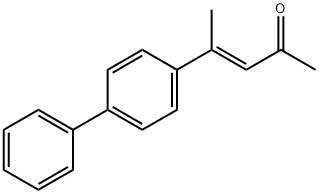
Xenipentone
- CAS No.
- 55845-78-8
- Chemical Name:
- Xenipentone
- Synonyms
- RP 4;RP48-482;RP-48-482;RP 48-482;Xenipentone;(E)-4-(4-Biphenylyl)-3-penten-2-one;(E)-4-(4-phenylphenyl)pent-3-en-2-one;3-Penten-2-one, 4-[1,1'-biphenyl]-4-yl-, (3E)-
- CBNumber:
- CB21180077
- Molecular Formula:
- C17H16O
- Molecular Weight:
- 236.31
- MDL Number:
- MFCD00869119
- MOL File:
- 55845-78-8.mol
| Boiling point | 394.1±22.0 °C(Predicted) |
|---|---|
| Density | 1.031±0.06 g/cm3(Predicted) |
| FDA UNII | GP581VM303 |
Xenipentone Chemical Properties,Uses,Production
Originator
Xenipentone ,ZYF Pharm Chemical
Manufacturing Process
a). 3-(4-Biphenylyl)-2-butenoic acid ethyl ester:
6.45 g (0.1 mole) of activated zinc metal (20 mesh) is placed in a flask fitted
with a septum inlet and a magnetic stirrer. The system is maintained under a
nitrogen atmosphere and kept at a temperature of 25°C. A solution of 19.6 g
(0.1 mole) of 4-acetylbiphenyl in 75 ml of dry tetrahydrofuran and 75 ml of
trimethyl borate (distilled from calcium hydride) is injected and the mixture
stirred. 11.1 ml (0.1 mole) of freshly distilled ethyl bromoacetate is injected in
one shot and the mixture stirred at 25°C for 12 hours. A mixture of 25 ml of
concentrated ammonium hydroxide and 75 ml of glycerin is added, and the
aqueous phase is separated and extracted thrice with 25 ml portions of diethyl
ether. The combined organic extracts are dried over anhydrous magnesium
sulphate and the diethylether removed on a rotary evaporator, the residue is
vacuum distilled and the fraction distilling at 0.125 mm at 171-172°C is
collected. Recrystallisation of 3-(4-biphenylyl)-2-butenoic acid ethyl ester from
petroleum ether yields the heading compound, substantially in trans form.
b). 3-(4-Biphenylyl)-2-butenoic acid:
The 3-(4-biphenylyl)-2-butenoic acid ethyl ester is mixed with 6 g of 85%
potassium hydroxide in 100 ml of aqueous ethanol and the resulting mixture
heated on a steam bath for 30 min. The mixture is then cooled, poured into
ice and extracted twice with 25 ml portions of diethylether. The aqueous
phase is filtered over Celite and the filtrate acidified with 2 N hydrochloric acid to pH 4 and cooled. The resulting precipitate is filtered, washed with ether, air
dried with suction and then dried under high vacuum at 50°C to yield the 3-
(4-biphenylyl)-2-butenoic acid substantially in trans form.
c). 3-(4-Biphenylyl)-2-butenoic acid chloride:
The crude 3-(4-biphenylyl)-2-butenoic acid is dissolved in 200 ml of dry
tetrahydrofuran and 4 ml (0.055 mole) of thionyl chloride is added. The
solution is refluxed under a nitrogen atmosphere for 3 hours and the solvent
and excess thionyl chloride then distilled off. The resulting residue is flash
distilled in a microdistillation apparatus at 145-153°C/0.075 mm to yield the
3-(4-biphenylyl)-2-butenoic acid chloride, substantially in trans form.
d). 2-(p-Biphenylyl-2-pentene-4-one: 10.0 g. (0.039 mole) of crude 3-(4-
biphenylyl)-2-butenoic acid chloride, is dissolved in 200 ml of dry
tetrahydrofuran. The solution placed in a 500 ml round bottom flask fitted
with a septum inlet and magnetic stirrer, and held under a nitrogen
atmosphere. The solution is cooled to -30°C in a dry ice/isopropanol bath and
19.5 ml (0.039 mole) of a commercial 2 M methyl magnesium bromide
solution in dry toluene is added, dropwise, over 30 min. After the addition is
complete, the mixture is allowed to warm to room temperature and then
stirred for 1 hour. The reaction is quenched by the addition of 20 ml of
saturated ammonium chloride solution and the organic layer is separated. The
aqueous layer is extracted twice with 20 ml portions of ether and the
combined organic extracts are then dried over anhydrous magnesium sulphate
and evaporated to yield the heading compound, m.p. 130° to 133°C, after
recrystallization from petroleum ether.
Therapeutic Function
Antiinflammatory
Xenipentone Preparation Products And Raw materials
Raw materials
Preparation Products
Xenipentone Suppliers
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| TargetMol Chemicals Inc. | +1-781-999-5354 | support@targetmol.com | United States | 19973 | 58 |
| Supplier | Advantage |
|---|---|
| TargetMol Chemicals Inc. | 58 |