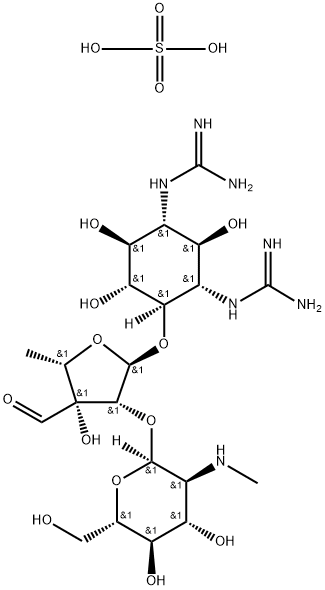
Streptomycin sulfate
- CAS No.
- 3810-74-0
- Chemical Name:
- Streptomycin sulfate
- Synonyms
- STREPTOMYCIN SULPHATE;Strepto;StreptoMycin sulfat;Streptomycin Sulphate Sterile;Estreptomicina;STREPTOMYCINI SULFAS;STREPTOMYCIN SULFATE SALT;STREPTOMYCIN SESQUISULFATE;Streptomycin Sulfate Sterile;as-15
- CBNumber:
- CB4290144
- Molecular Formula:
- C21H41N7O16S
- Molecular Weight:
- 679.65
- MDL Number:
- MFCD00037023
- MOL File:
- 3810-74-0.mol
- MSDS File:
- SDS
| Melting point | >185°C (dec.) |
|---|---|
| alpha | -79~-88°(20℃/D)(c=1,H2O)(calculated on the dried basis) |
| Density | 1.2302 (rough estimate) |
| refractive index | -85 ° (C=1, H2O) |
| storage temp. | 2-8°C |
| solubility | H2O: 0.1 g/mL, clear |
| form | powder |
| color | white to off-white |
| PH | pH (100g/l, 25℃) : 4.5~7.0 |
| Water Solubility | >=0.01 g/100 mL at 18 ºC |
| Merck | 14,8826 |
| BRN | 3894995 |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| InChIKey | QTENRWWVYAAPBI-YZTFXSNBSA-N |
| LogP | -1.523 (est) |
| CAS DataBase Reference | 3810-74-0 |
| FDA UNII | CW25IKJ202 |
| Proposition 65 List | Streptomycin Sulfate |
| Pesticides Freedom of Information Act (FOIA) | Streptomycin sulfate |
| EPA Substance Registry System | Streptomycin sulfate (3810-74-0) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS07,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H361fd | |||||||||
| Precautionary statements | P201-P202-P264-P270-P301+P312-P308+P313 | |||||||||
| Hazard Codes | Xn,Xi | |||||||||
| Risk Statements | 22-36/37/38-63-42/43 | |||||||||
| Safety Statements | 26-36-36/37-45-22-60 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | WK4990000 | |||||||||
| F | 3 | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 29412000 | |||||||||
| NFPA 704 |
|
Streptomycin sulfate price More Price(51)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | Y0002232 | Streptomycin sulfate for identification European Pharmacopoeia (EP) Reference Standard | 3810-74-0 | Y0002232 | $156 | 2024-03-01 | Buy |
| Sigma-Aldrich | PHR2687 | Streptomycin Sulfate Pharmaceutical Secondary Standard; Certified Reference Material, certified reference material, pharmaceutical secondary standard, pkg of 500?mg | 3810-74-0 | 500MG | $226 | 2024-03-01 | Buy |
| Sigma-Aldrich | 5711 | Streptomycin Sulfate, Streptomyces sp. - CAS 3810-74-0 - Calbiochem | 3810-74-0 | 100g | $99.4 | 2024-03-01 | Buy |
| Sigma-Aldrich | 46754 | Streptomycin sesquisulfate hydrate VETRANAL | 3810-74-0 | 250mg | $63.7 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1623003 | Streptomycin sulfate United States Pharmacopeia (USP) Reference Standard | 3810-74-0 | 200mg | $436 | 2024-03-01 | Buy |
Streptomycin sulfate Chemical Properties,Uses,Production
Streptomycin sulfate
Streptomycin sulfate is the sulfate form of streptomycin. Streptomycin class antibiotics are produced by the soil-living Actinomyces Streptomyces. Streptomycin is the first emerging drug which has been proven to be an effective chemotherapy drug in treating tuberculosis. This product must be injected intramuscularly which makes it be limited for long-term application. Streptomycin, obtained from Streptomyces, is currently the most effective and also the least toxic antibiotics for application outside the gastrointestinal tract.This product is a kind of fungicides which may be due to its direct effect on the bacterial ribosome, and the resulting effective inhibition of protein synthesis. It is mainly used for treating extracellular (including the cavity) Mycobacterium tuberculosis.
Streptomycin sulfate has a strong antibacterial effect against Mycobacterium tuberculosis but has only a mild antibacterial effect on most Gram-positive cocci (such as various Streptococcus) and tuberculosis (such as Pseudomonas aeruginosa, anaerobic). However, it still has a strong antibacterial effect on many Gram negative bacteria. The product is effective in treating a variety of skin tuberculosis with an effect of inhibiting the proliferation of Mycobacterium tuberculosis and the production of toxin. Moreover, at high concentrations (> 0.4 μg/mL), it has bactericidal effect. However, Mycobacterium tuberculosis can rapidly becomes resistance to streptomycin, and thus should be combined with other anti-TB drugs. Combing streptomycin sulfate with another approved parenteral-applied efficient fungicide, isoniazid can immediately yield a significant effect on treating susceptible bacteria. It often has resurrecteding effect in some critical situations.
Using streptomycin sulfate for treatment during the first few weeks or months of treatment can achieve the largest efficacy perhaps because of parenteral application of the strain. Serum concentrations can increase rapidly thus seems to further enhance the oral administration efficacy. Even for efficient drugs like ethambutol and isoniazid can also benefit from the compatibility of this product.
Clinical studies have shown: the four-drug therapy including streptomycin, rifampin, isoniazid and pyrazinamide can produce the strongest bactericidal effect. Treating with this therapy for two months and further treatment with isoniazid and rifampicin for 4 months can yield an efficacy which is comparable to efficacy of any other kinds of therapy being used for treating susceptible bacterial infections (Algeria Study group/British Medical Research Council, 1984; Snider, etc., 1984B, 1986).
Physical and Chemical Properties
White or white-like powder, odorless, slightly bitter taste, hygroscopic, soluble in water, easily soluble in water and relatively stable, insoluble in ethanol or chloroform, be stable under dry condition, can be sealed from light for more than four years with efficacy unchanged, the aqueous solution can be stored at room temperature pH3~7 for 2 to 4 weeks, in the case of acid, alkali, oxidizing agents, it is easily damaged and lose effect.
Pharmacological effects
Streptomycin sulfate has antibacterial effects on most Gram-positive and Gram-negative bacteria especially has a strong antibacterial effects on a variety of Gram-negative bacteria such as Escherichia.coli, Salmonella, Pasteurella, Brucella, Klebsiella pneumoniae, Shigella, gas production Bacillus, Burkholderia pseudomallei. This product is a first-choice for anti-tuberculosis drug which has a relative strong effect on Mycobacterium tuberculosis. It has a strong inhibitory effect at low concentrations and has bactericidal effect at high concentrations. However, due to that only 10% can penetrate cells, so it is ineffective to kill intracellular Mycobacterium tuberculosis but can only kill extracellular Mycobacterium tuberculosis. It has a stronger bactericidal effect on breeding bacteria than bacteria in stationary phase. Being different from penicillin, its bactericidal rate is proportional to the concentration. Its antibacterial activity is strongest at pH 7.8 but is greatly reduced when pH drop to below 6.0.
Agricultural antibiotic streptomycin sulfate belongs to antibiotics-class fungicides which can be used for preventing and treating a variety of bacterial disease for crops; it also has some prevention and treatment effect on some fungal diseases. According to our pesticide toxicity grading standards, streptomycin belong to low toxicity fungicides. For original drug with acute oral administration, rat LD50> 9 000mg /kg body weight. It can cause allergic skin reaction.
Pharmacodynamics
After streptomycin sulfate enter into the body tissue through the cell membranes of pathogenic microorganisms, it binds to specific receptor proteins of the 30S ribosome subunit in pathogenic microorganisms and interfere with the formation of the protein translation initiation complex between the mRNA and the 30S subunit which cause DNA errors as well as causing the completion with paired amino acids from unpaired amino acids rivalry, resulting in the synthesis of non-functional protein, so that pathogenic microorganisms polysome are dissociated, causing the ultimate failure of protein synthesis, and further causing the death of pathogenic microorganisms.
Pharmacokinetics
Drug through intramuscular injection is well absorbed. It is widely distributed in the body's organs and tissues, but can rarely penetrate through the blood-brain barrier for entering into the brain tissue. Pharmaceuticals are primarily located in the extracellular fluid and can enter the bile, chest, ascites, tuberculous abscess, and cheese-like tissue; high drug concentrations can be detected in urine. It can also enter into the fetal tissue through the placenta. It only has a protein binding rate as low as 20% to 30%. Upon intramuscular injection of 1g drugs, plasma concentration reaches peak 25~50μg/mL in 0.5~1.5 h with a half-life of 2.5~3h. Within 24 h, 80%~98% of the administered dose is excreted out from the urine with about 1% being discharged from the bile, and also a very small amount discharged from the saliva and breast milk.
Clinical application
This product is effective against Gram-positive bacteria and gram-negative bacteria. But it is less effective than penicillin G in treating Gram-positive bacteria such as Streptococcus pneumoniae bacteria, and has no effects on Rickettsia, protozoa, viruses and fungi; It has a good effect on treating Mycobacterium tuberculosis and many kinds of gram-negative bacteria such as Escherichia.coli, Brucella, gas, influenza bacillus, Shigella, and Yersinia pestis. The main clinical application is used for the treatment of Mycobacterium tuberculosis induced diseases cuh as tuberculosis, bone tuberculosis, lymphatic, intestinal tuberculosis, tuberculous meningitis and meningitis caused by susceptible strains, pneumonia, sepsis, empyema, pleurisy, peritonitis, etc., It can also be used for treating infection of urinary tract and respiration system caused by susceptible strains.
Drug Interactions
1. Combination of Streptomycin and penicillin or cephalosporins has synergistic effect.
2. its antibacterial activity can be enhanced in alkaline environment, and being used in combination with basic drugs (such as sodium bicarbonate) can further enhance the antimicrobial potency.
3. Cations such as Ca2 +, Mg2 +, Na+, NH4+, K+ inhibit the antibacterial activity of the product.
4. Combination of streptomycin sulfate with cephalosporins, erythromycin can enhance the ototoxicity of this class of drugs.
The above information is edited by the Chemicalbook of Dai Xiongfeng.
Side effects
The adverse reactions of streptomycin are both more and more serious than penicillin. Ototoxicity is the most serious adverse reactions of streptomycin. This product can often badly affect lthe vestibular branches of the listening nerve, causing nausea, vomiting and dizziness. The incidence of ototoxicity is directly related to the dose and duration of treatment. Old age and renal dysfunction is the main cause. Hearing loss may occurs and often permanently in some severe cases. The neurotoxicity of this product can cause respiratory paralysis (due to neuromuscular blockade). Patients who have just been subject to anesthesia and muscle relaxants are especially prone to get these adverse reactions.
Streptomycin-induced renal toxicity only occasionally occurs, but if the patents who originally has a reduced kidney function or has been applied with kidney-toxic drugs at the same time can increase the incidence. Thereby they should avoid the simultaneous or sequential application of other neurotoxic or nephrotoxic drugs.
Since for over 65 years old men, the ototoxicity and renal toxicity are quite common, therefore for this age group, streptomycin should try to be avoided. For all the cases, the patients should be subject to basic and regular audiometric and vestibular caloric test.
In proper use, streptomycin toxicity rarely occurs with most patients being tolerated well. Individual patients can rapidly get transient headaches or discomfort after injection. The incidence of irrelevant paresthesias in the face (especially perioral) is about 15% in all the cases with sometimes also being accompanied by tingling hands.
During the first few weeks of treatment of streptomycin, allergic reactions occasionally occur but not as frequently as amino salicylic acid and also generally not as seriously as amino salicylic acid and isoniazid. Anaphylactic shock and hematological reactions such as increased acidity cells, agranulocytosis, and aplastic anemia have also been sporadically reported before.
The teratogenicity effect of this product has been already confirmed by animal experiments. This product is not suitable for use in the early three months of pregnancy as well as the later-half period of pregnancy. Moreover, the total dose should not be allowed to exceed 20g in order to prevent congenital deafness.
Mother who applied this drug can have trace amount entering into milk when serum concentration have achieved therapeutic levels.
Precautions
Allergy history must be asked before administration. For patients with no history of allergy, skin test must be done in order to confirm negative result before administration. Upon skin test, take 00mg/0.1ml of the dilution solution for intradermal injection and observe for 15min. Patients with poor liver function should be cautious when using. Injection should not be mixed with the salt of penicillin G which will cause the change of solution pH during mixing, resulting in a reduced potency of penicillin.
During the course of medication, when adverse reactions such as dizziness, hearing loss, and tinnitus occur, stop using the drug immediately and reduce the dose if necessary. Infants should take with caution.
For elderly people, application of drugs can cause many kinds of toxic reactions. Thereby, renal function should be monitored in the course of treatment; It can cause the overestimation of the determined value of alanine aminotransferase, aspartate aminotransferase, serum bilirubin concentration and serum concentration of lactate dehydrogenase as well as the determined value of calcium, magnesium, potassium, sodium; Pay attention to the electrical diagram and the renal function of the patients.
Uses
It is used for treating tuberculosis, Brinell and non-hemolytic hammer bacillus caused infectious endocarditis, plague and tularemia, Haemophilus influenzae and gram-negative bacilli caused infections.
Description
Streptomycin was found in the culture broth of Streptomyces griseus by S. A. Waksman of Rutgers University in 1944; it was the second antibiotic introduced clinically (after penicillin). This drug is a water soluble, basic substance having an aminoglycoside structure and showing strong activity against a wide range of gram-positive and gram-negative bacteria including Mycobacterium. Streptomycin is the first choice among antituberculotic antibiotics and has been used for the therapy of Spirochaeta and Treponema infections.
Chemical Properties
White to off-white powder
Uses
Triacetine
Uses
Antibiotic substance produced by aerobic fermentation. Antibacterial (tuberculostatic).
Uses
Streptomycin sulfate is a sulfate salt of streptomycin that is a protein synthesis inhibitor. Streptomycin is an antibiotic drug, the first of a class of drugs called aminoglycosides to be discovered, and was the first antibiotic remedy for tuberculosis.
Definition
ChEBI: Streptomycin sulfate is an aminoglycoside sulfate salt. It is functionally related to a streptomycin.
brand name
Strycin (Bristol-Myers Squibb).
General Description
An antibacterial. White to light gray or pale buff powder with faint amine-like odor.
Air & Water Reactions
Hygroscopic. Water soluble.
Reactivity Profile
Streptomycin sulfate is a medicinal amino saccharide.
Health Hazard
SYMPTOMS: Symptoms of exposure to Streptomycin sulfate include skin rashes, eosinophilia, blood dyscrasias, stomatitis, anaphylactic shock, pruritus, scaling, and fever.
Fire Hazard
Flash point data for Streptomycin sulfate are not available, however Streptomycin sulfate is probably combustible.
Clinical Use
Streptomycin sulfate is a white, odorless powder that is hygroscopicbut stable toward light and air. It is freely solublein water, forming solutions that are slightly acidic or nearlyneutral. It is very slightly soluble in alcohol and is insolublein most other organic solvents. Acid hydrolysis yields streptidineand streptobiosamine, the compound that is a combinationof L-streptose and N-methyl-L-glucosamine.
Streptomycin acts as a triacidic base through the effectof its two strongly basic guanidino groups and the moreweakly basic methylamino group. Aqueous solutions maybe stored at room temperature for 1 week without any lossof potency, but they are most stable if the pH is between 4.5and 7.0. The solutions decompose if sterilized by heating,so sterile solutions are prepared by adding sterile distilledwater to the sterile powder. The early salts of streptomycincontained impurities that were difficult to remove andcaused a histamine-like reaction. By forming a complexwith calcium chloride, it was possible to free the streptomycinfrom these impurities and to obtain a product thatwas generally well tolerated.
The organism that produces streptomycin, S. griseus, alsoproduces several other antibiotic compounds: hydroxystreptomycin,mannisidostreptomycin, and cycloheximide (q.v.).Of these, only cycloheximide has achieved importance as amedicinally useful substance. The term streptomycin A hasbeen used to refer to what is commonly called streptomycin,and mannisidostreptomycin has been called streptomycin B.Hydroxystreptomycin differs from streptomycin in having ahydroxyl group in place of one of the hydrogen atoms of thestreptose methyl group. Mannisidostreptomycin has a mannoseresidue attached in glycosidic linkage through the hydroxylgroup at C-4 of the N-methyl-L-glucosamine moiety.The work of Dyer et al. to establish the stereochemicalstructure of streptomycin has been completed, and confirmedwith the total synthesis of streptomycin and dihydrostreptomycinby Japanese scientists.
Safety Profile
Poison by ingestion and intravenous routes. Moderately toxic by subcutaneous route. An experimental teratogen. Human systemic effects by intraperitoneal route: flaccid paralysis without anesthesia, motor activity changes and pulmonary changes. Experimental reproductive effects. Mutation data reported. When heated to decomposition it emits very toxic fumes of NOx and SOx. See also STREPTOMYCIN and SULFATES.
Purification Methods
The sulfate is recrystallised from H2O/EtOH, washed with a little EtOH, Et2O and dried in a vacuum. [UV and IR: Grove & Randall Antibiotics Monographs 2 163 1855, Heuser et al. J Am Chem Soc 75 4013 1953, Kuehl et al. J Am Chem Soc 68 1460 1946, Regna et al. J Biol Chem 165 631 1946.] During protein synthesis it inhibits initiation and causes misreading of mRNA [Zierhut et al. Eur J Biochem 98 577 1979, Chandra & Gray Methods Enzymol 184 70 1990]. [Beilstein 18/11 V 82.]
Streptomycin sulfate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| shandong perfect biotechnology co.ltd | +86-53169958659; +8618596095638 | sales@sdperfect.com | China | 294 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 | deasea125996@gmail.com | China | 2503 | 58 |
| Hebei Jingbo New Material Technology Co., Ltd | +8619931165850 | hbjbtech@163.com | China | 1000 | 58 |
| Shanghai Affida new material science and technology center | +undefined15081010295 | 2691956269@qq.com | China | 359 | 58 |
| Hebei Saisier Technology Co., LTD | +86-18400010335 +86-13102810335 | admin@hbsaisier.cn | China | 747 | 58 |
| Shaanxi TNJONE Pharmaceutical Co., Ltd | +8618740459177 | sarah@tnjone.com | China | 893 | 58 |
| Ouhuang Engineering Materials (Hubei) Co., Ltd | +8617702722807 | admin@hbouhuang.com | China | 2259 | 58 |
| Shanghai Daken Advanced Materials Co.,Ltd | +86-371-66670886 | info@dakenam.com | China | 15928 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21691 | 55 |
| Hangzhou FandaChem Co.,Ltd. | 008657128800458; +8615858145714 | fandachem@gmail.com | China | 9348 | 55 |
Related articles
- Streptomycin Sulfate: Biological Activities, Efficacy in Humans and Safety
- Streptomycin sulfate is an aminoglycoside antibiotic used to treat Gram-negative infections, including tuberculosis. It has po....
- Jan 17,2024
- Introduction and Preparation of Streptomycin Sulfate
- Streptomycin sulfate is the sulfate salt form of?streptomycin, an aminoglycoside antibiotic derived from Streptomyces griseus ....
- Nov 18,2022
View Lastest Price from Streptomycin sulfate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-04-26 | Streptomycin sulfate
3810-74-0
|
US $0.00 / kg | 25kg | 720IU/mg | 10tons | Henan Suikang Pharmaceutical Co.,Ltd. | |
 |
2024-04-24 | Steptomycin sulfate
3810-74-0
|
US $15.00 / kg | 1kg | 99.912% | 10ton | Ouhuang Engineering Materials (Hubei) Co., Ltd | |
 |
2024-04-24 | Streptomycin Sulphate
3810-74-0
|
US $0.00 / kg | 1kg | 99% | 1000kgs | Shaanxi TNJONE Pharmaceutical Co., Ltd |
-

- Streptomycin sulfate
3810-74-0
- US $0.00 / kg
- 720IU/mg
- Henan Suikang Pharmaceutical Co.,Ltd.
-

- Steptomycin sulfate
3810-74-0
- US $15.00 / kg
- 99.912%
- Ouhuang Engineering Materials (Hubei) Co., Ltd
-

- Streptomycin Sulphate
3810-74-0
- US $0.00 / kg
- 99%
- Shaanxi TNJONE Pharmaceutical Co., Ltd
3810-74-0(Streptomycin sulfate )Related Search:
1of4