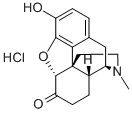
Hydromorphone hydrochloride
- CAS No.
- 71-68-1
- Chemical Name:
- Hydromorphone hydrochloride
- Synonyms
- HYDROMORPHONE HCL;dilaudidhydrochloride;Hydromorphine hydrochloride;HYDROMORPHONE HYDROCHLORIDE;XHILEZUETWRSHC-NRGUFEMZSA-N;HYDROMORPHONEHYDROCHLORIDE,USP;dihydromorphinonehydrochloride;Hydromorphone hydrochloride salt;Morphinone, dihydro-, hydrochloride;hydromorphone hydrochloride solution
- CBNumber:
- CB7250921
- Molecular Formula:
- C17H20ClNO3
- Molecular Weight:
- 321.8
- MDL Number:
- MFCD03427563
- MOL File:
- 71-68-1.mol
| Melting point | >280°C (dec.) |
|---|---|
| alpha | D25 -133° |
| Density | 1.1451 (rough estimate) |
| refractive index | 1.6470 (estimate) |
| storage temp. | 2-8°C |
| solubility | H2O: >10 mg/mL |
| form | A crystalline solid |
| NCI Dictionary of Cancer Terms | hydromorphone hydrochloride |
| FDA UNII | L960UP2KRW |
| NCI Drug Dictionary | hydromorphone hydrochloride |
| EPA Substance Registry System | Morphinan-6-one, 4,5-epoxy-3-hydroxy-17-methyl-, hydrochloride (1:1), (5.alpha.) (71-68-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H336 | |||||||||
| Precautionary statements | P301+P312+P330 | |||||||||
| Hazard Codes | Xn | |||||||||
| Risk Statements | 22 | |||||||||
| Safety Statements | 16-36/37-45 | |||||||||
| RIDADR | UN 1230 3/PG 2 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | QD2625000 | |||||||||
| HazardClass | 6.1(a) | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 2939110000 | |||||||||
| Toxicity | LD50 in mice (mg/kg): 61-96 i.v. (Buchwald, Eadie) | |||||||||
| NFPA 704 |
|
Hydromorphone hydrochloride Chemical Properties,Uses,Production
Description
Hydromorphone hydrochloride is a pure opioid agonist with the principal therapeutic activity of analgesia. It has an analgesic potency approximately two to eight times greater than that of morphine and has a rapid onset of action. A significant feature of the analgesia is that it can occur without loss of consciousness.
Chemical Properties
White Solid
Uses
Analgesic (narcotic). Preparation by electrolytic reduction of Morphine. Controlled substance (opiate).
Biochem/physiol Actions
Narcotic opiate analgesic; μ opioid receptor agonist.
Clinical Use
Hydromorphone is a potent μ agonist (eight times greater than morphine) that is used to treat severe pain. It is available in intramuscular, intravenous, subcutaneous, oral, and rectal dosage forms. Like all strong μ agonists, hydromorphone is addicting and is a Schedule II drug. Hydromorphone has an oral:parenteral potency ratio of 5:1. The plasma half-lives after parenteral and oral dosage are 2.5 and 4 hours, respectively.
Safety Profile
Poison by subcutaneous and intravenous routes. Experimental teratogenic effects. A powerful analgesic. When heated to decomposition it emits very toxic fumes of NOx and HCl. See also MORPHINE.
Drug interactions
Potentially hazardous interactions with other drugs
Alcohol: can cause dose dumping with sustained
release preparations.
Analgesics: possible opioid withdrawal effects with
buprenorphine and pentazocine.
Antidepressants: possible CNS excitation or
depression with MAOIs - avoid concomitant use
and for 2 weeks after stopping MAOI; possible
CNS excitation or depression with moclobemide;
increased sedative effects with tricyclics.
Antihistamines: increased sedative effects with
sedating antihistamines.
Antipsychotics: enhanced hypotensive and sedative
effects.
Dopaminergics: avoid with selegiline.
Nalmefene: avoid concomitant use.
Sodium oxybate: enhanced effect of sodium oxybate
- avoid.
Metabolism
Hydromorphone undergoes extensive firstpass metabolism. It is extensively metabolised by glucuronidation in the liver and excreted in the urine mainly as conjugated hydromorphone, dihydroisomorphine, and dihydromorphine.