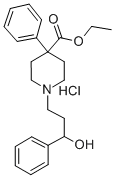
phenoperidine hydrochloride
- CAS No.
- 3627-49-4
- Chemical Name:
- phenoperidine hydrochloride
- Synonyms
- Lealgin;HGEAGOBQOIMDQV-UHFFFAOYSA-N;phenoperidine hydrochloride;3-(4-Carbethoxy-4-phenylpiperidino)-1-phenyl-1-propanol Hydrochloride;1-(3-Hydroxy-3-phenylpropyl)-4-phenylisonipecotic Acid Ethyl Ester Hydrochloride;1-(3-Hydroxy-3-phenylpropyl)-4-phenyl-4-piperidinecarboxylic Acid Ethyl Ester Hydrochloride
- CBNumber:
- CB9913457
- Molecular Formula:
- C23H29NO3.ClH
- Molecular Weight:
- 403.947
- MDL Number:
- MOL File:
- 3627-49-4.mol
| Melting point | 200-202° |
|---|---|
| FDA UNII | 1D9C1OK7KZ |
phenoperidine hydrochloride Chemical Properties,Uses,Production
Originator
Operidine ,Janssen ,US ,1965
Uses
Analgesic (narcotic). Controlled substance (opiate).
Manufacturing Process
The starting materials for the overall process are phenylacetonitrile with bischloroethyl toluene sulfonyl amide. These react to give a product which hydrolyzes to normeperidine (4-carboethoxy-4-phenylpiperidine). Condensation of that material with benzoylethylene gives the ketone: β-(4carboethoxy-4-phenylpiperidino)propiophenone.
A reaction mixture was prepared containing 4 grams of β-(4-carboethoxy-4phenylpiperidino)-propiophenone hydrochloride, 100 ml of methanol and about 0.5 gram of platinum oxide catalyst. The mixture was placed in a low pressure hydrogenation apparatus and was hydrogenated at a temperature of about 27°C and a pressure of about 3.5 atmospheres of hydrogen to convert the keto group of the β-(4-carboethoxy-4-phenylpiperidino)-propiophenone to a hydroxy group, and to form 3-(4-carboethoxy-4-phenylpiperidino)-1-phenyl-1propanol hydrochloride. After the hydrogenation was complete, the catalyst was separated from the reaction mixture by filtration, and the filtrate was evaporated to dryness in vacuo leaving a residue containing 3-(4carboethoxy-4-phenylpiperidino)-1-phenyl-l-propanol hydrochloride. The residue was digested with ethyl acetate thereby causing 3-(4-carboethoxy-4phenylpiperidino)-1-phenyl-1-propanol hydrochloride to crystallize. This compound melted at about 188°-189°C after being recrystallized three times from an ethyl acetate-methanol solvent mixture, according to US Patent 2,951,080.
Therapeutic Function
Analgesic
Safety Profile
A poison by intraperitoneal route. When heated to decomposition it emits toxic vapors of NOx, HCl, and Clí.