ドデセン-1 LAS 化学特性,用途語,生産方法
外観
無色澄明の液体
定義
本品は、ドデシルベンゼンスルホン酸(*)のナトリウム塩であり、次の化学式で表される。
参照表示名称:ドデシルベンゼンスルホン酸
溶解性
水に溶けやすい。
用途
陰イオン界面活性剤定量標準品。
化粧品の成分用途
洗浄剤
使用上の注意
アルゴン封入
説明
Sodium dodecyl benzene sulfonate is a series of organic compounds with the formula C12H25C6H4SO3Na. It is a colourless salt with useful properties as a surfactant. It is usually produced as a mixture of related sulfonates. It is a major component of laundry detergent.
化学的特性
white or light yellow flakes
使用
A surfactant used in proteomics research.
一般的な説明
Sodium dodecylbenzenesulfonate is a white to light yellow flakes, granules or powder. Sodium dodecylbenzenesulphonate is soluble in water. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Sodium dodecylbenzenesulphonate is used as a synthetic detergent.
空気と水の反応
Sodium dodecylbenzenesulphonate is soluble in water.
反応プロフィール
SODIUM DODECYLBENZENESULFONATE is incompatible with strong oxidizers.
健康ハザード
Minor skin and eye irritant. INGESTION: May cause vomiting, diarrhea, and intestinal distension.
工業用途
These frothers are mixtures of alcohols containing 6–8 carbon atoms. They were at one
time marketed by DuPont and they are tailored frothers for specific ore types. The bestknown
frother from this group is methyl isobutyl carbinol (MIBC) and 2-ethyl hexanol.
Aliphatic alcohol frothers are used as mixtures of different carbon lengths and as a
mixture of hydrocarbon oils.
安全性プロファイル
Poison by intravenous
route. Moderately toxic by ingestion. A skin and
severe eye irritant. When heated to decomposiuon
it emits tomc fumes of NazO. See also
SULFONATES
合成
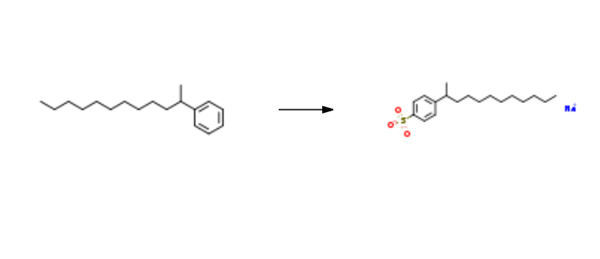
Sodium dodecylbenzenesulphonate is synthesised using 2-phenyldodecane as raw material by chemical reaction. The specific synthesis steps are as follows:
To a 5 L round bottom flask equipped with mechanical stirrer and an addition funnel was added octadecane and 2-phenyldodecane mixture ( 1.73 kg, 28%> 2-phenyldodecane). The reaction mixture was sparged with argon, warmed to 35°C and 1.25 weight % of oleum (632 g, 1.58 mol) was added dropwise, via addition funnel, to reaction mixture. The reaction mixture was stirred for 1.5 hours at room temperature. Upon completion, the reaction mixture was heated to 50°C and transferred to a separatory funnel and allowed to separate. The bottom layer was added slowly to a stirred solution of 15%> NaOH (aq) (2 L) at 10°C. Upon complete addition the resulting suspension was stirred for an additional 60 minutes. The solid was subsequently isolated by filtration and washed twice with ice-cold water. The solids were air dried for 16 hours and vacuum dried at 80°C to yield sodium, 4-(dodecan-2-yl) benzenesulfonate (555 g, 80.8% yield, 98.5% purity). lH NMR (400 MHz, (CD3)2S0/CDC13) δ 0.84 (t, J = 7.0 Hz, 3H), 0.95- 1.38 (m, 19H), 1.51 (pquart, J = 7.3 Hz, 2H), 2.65 (psext, J = 7.0 Hz, 1 H), 7.1 1 (d, J = 7.6 Hz, 2H), 7.56 (d, J = 8.4 Hz, 2H). 13C NMR (101 MHz, (CD3)2S0/CDC13) δ 13.8, 22.0, 22.1 , 27.0, 28.9, 28.9, 28.9, 28.9, 31.2, 37.6, 38.8, 125.4, 125.8, 145.3, 147.7.
純化方法
It crystallises from propan-2-ol or H2O. [Gray et al. J Org Chem 20 515 1955, Beilstein 11 IV 514.]
ドデセン-1 LAS 上流と下流の製品情報
原材料
準備製品