아파티닙 C화학적 특성, 용도, 생산
용도
Afatinib is a tyrosine kinase receptor inhibitor that is used to treat metastatic (cancer that has already spread) non-small cell lung cancer (NSCLC) that has certain types of abnormal epidermal growth factor receptor (EGFR) genes in patients who have not received any treatments for cancer that has already spread to other parts of the body. This medicine is also used to treat patients with metastatic squamous NSCLC who have received medicines containing platinum but did not work well.
Indications
The collection of ibrutinib (Imbruvica(R), Pharmacyclics Inc.), afatinib, and osimertinib represents the small, yet expanding, group of covalent SMKIs. Ibrutinib is a non-receptor Bruton’s tyrosine kinase inhibitor approved for the treatment of relapsed chronic lymphocytic leukemia. Afatinib, approved for NSCLC in 2013 and squamous NSCLC in 2016, is a second-generation irreversible EGFR inhibitor that targets wild-type EGFR, the mutant T790M EGFR, and HER2. Osimertinib (AZD9291), which was approved by FDA in November 2015, is a third-generation irreversible EGFR inhibitor that selectively targets the mutant T790M EGFR. Rociletinib, which shares a high degree of structural similarity with that of osimertinib, is a promising covalent EGFR inhibitor developed by Clovis Oncology aimed for the treatment of patients with EGFR T790M-mutated NSCLC, until the company terminated its development in May 2016 following a negative vote fromthe FDA’sOncologic Drugs Advisory Committee.
정의
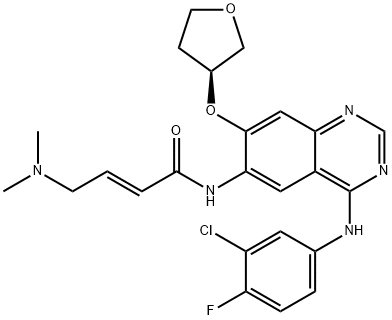
ChEBI: Afatinib is a quinazoline compound having a 3-chloro-4-fluoroanilino group at the 4-position, a 4-dimethylamino-trans-but-2-enamido group at the 6-position, and an (S)-tetrahydrofuran-3-yloxy group at the 7-position. Used (as its dimaleate salt) for the first-line treatment of patients with metastatic non-small cell lung cancer. It has a role as a tyrosine kinase inhibitor and an antineoplastic agent. It is a member of quinazolines, a member of furans, an organofluorine compound, an enamide, an aromatic ether, a tertiary amino compound, a member of monochlorobenzenes and a secondary carboxamide.
효소 저해제
This oral quinazoline derivative and EGFR/HER2-directed protein kinase inhibitor (FW = 485.94; CASs = 439081-18-2 (free base), 936631-70-8 (maleic acid salt), 1254955-21-9 (HCl salt); Solubility (at 25°C): 197 mg/mL DMSO, 1 mg/mL Water), also known by its code name BIBW2992, its trade names Gilotrif? Tomtovok?, Tovok?, and its systematic name (S,E)-N-(4-(3-chloro-4-fluorophenyl-amino)-7-(tetrahydrofuran- 3-yloxy)quinazolin-6-yl)-4-(dimethylamino)but-2-enamide, irreversibly inactivates EGFRwt, EGFRL858R, EGFRL858R/T790M and HER2with IC50 values of 0.5 nM, 0.4 nM, 10 nM and 14 nM, respectively. Mode of Action: The irreversible binding of afatinib to HER2 inactivates its interactions with a preferred partner of EGFR, and blocking the HER2- EGFR heterodimerization reduces their intrinsic tyrosine kinase activities. Irreversible inhibitors (such as afatinib and dacomitinib) that target all ErbB family receptor tyrosine kinases are intended to confer sustained disease control in ErbB-dependent cancers. Because nearly all EGFRmutated patients eventually develop resistance to reversible EGFR-TKIs after a median of 14 months, tafatinib’s irreversible action is thought to be a promising feature of its mode of action. Pharmacokinetics: Afatinib’s PK profile is best described by a two-compartment disposition model, with first-order absorption and linear elimination. There was a slightly more than proportional increase in exposure with increasing dose, most likely due to dose-dependent relative bioavailability. For the therapeutic dose of 40 mg, the estimated apparent total clearance rate at steady state was 734 mL/min.
아파티닙 준비 용품 및 원자재
원자재
준비 용품