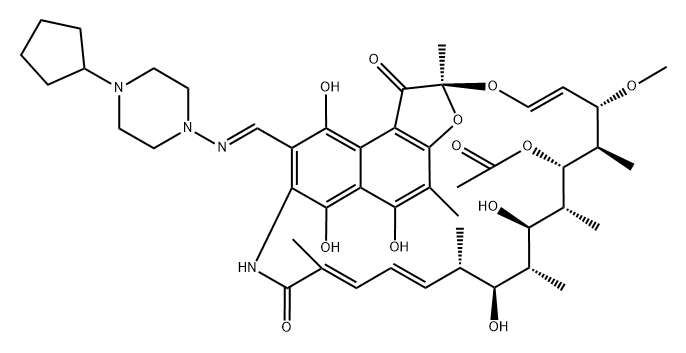
Rifapentine
|
|
Rifapentine 속성
- 녹는점
- 179-1800C
- 끓는 점
- 969.3±65.0 °C(Predicted)
- 밀도
- 1.35±0.1 g/cm3(Predicted)
- 저장 조건
- Sealed in dry,Store in freezer, under -20°C
- 용해도
- 메탄올: 용해성2mg/mL, 투명, 빨간색~적갈색
- 물리적 상태
- 가루
- 산도 계수 (pKa)
- 4.81±0.70(Predicted)
- 색상
- 빨간색
안전
- 위험 및 안전 성명
- 위험 및 사전주의 사항 (GHS)
| 위험품 표기 | Xi | ||
|---|---|---|---|
| 위험 카페고리 넘버 | 36/37/38 | ||
| 안전지침서 | 26 | ||
| WGK 독일 | 3 | ||
| RTECS 번호 | JQ0902000 | ||
| HS 번호 | 2941.90.3000 | ||
| 독성 | LD50 in mice (mg/kg): >2000 orally; 750 i.p. (Cricchio); LD50 in mice (mg/kg): 3300 orally; 710 i.p. (Aroli) |
Rifapentine C화학적 특성, 용도, 생산
개요
Rifapentine is a broad spectrum antibiotic highly active against Gram-positive bacteria and Neisseria gonorrhoea. It is reported to be 10 times more active than the structurally related rifampicin against Mycobacrerim fuberculosis. Unlike rifampicin the bioavailability of rifapentine is significantly increased when administered after a meal.화학적 성질
Crystalline Solid용도
Semi-synthetic rifamycin. Antibacterial (tuberculostatic)Indications
Rifapentine is an analogue of rifampin that is active against M. tuberculosis and M. avium. Rifapentine’s mechanism of action, cross-resistance, hepatic induction of P450 enzymes, drug interactions, and toxic profile are similar to those of rifampin. It has been used in the treatment of tuberculosis caused by rifampinsusceptible strains.Antimicrobial activity
Activity is similar to that of rifampicin, but it is more active against atypical mycobacteria, especially the M. avium complex (MIC <0.06–0.5 mg/L). It has good activity on staphylococci and streptococci (MIC 0.01–0.5 mg/L), L. monocytogenes and Brucella spp.; less against Enterococcus faecalis (MIC 1–4 mg/L). Bacteroides spp. are inhibited by 0.5–2 mg/L. Gram-negative cocci are susceptible and, although some Gram-negative bacilli are inhibited by 4–32 mg/L, most are resistant.위험도
Moderately toxic by ingestion.Pharmaceutical Applications
An analog of rifampicin in which a cyclopentyl group is substituted for a methyl group on the piperazine ring. It is available for oral administration.Mechanism of action
Because relapse and the emergence of resistant strains of bacteria are associated with poor patient compliance, reduced dosing is expected to increase compliance. Initial clinical studies actually showed that the relapse rates in patients treated with rifapentine (10%) were higher than those in the patients treated with RIF (5%). It was found that poor compliance with the nonrifamycin antituberculin agents was responsible for the increased relapse.Pharmacokinetics
Oral absorption:c. 70%Cmax600 mg oral :12 mg/L after 5 h
Plasma half-life:13 h
Volume of distribution:1.5 L/kg
Plasma protein binding:97%
absorption
The absolute oral bioavailability of rifapentine has not been determined. The relative bioavailability of capsules (with an oral solution as reference) is 70%. Food increases absorption: a 600 mg dose taken after a meal gives Cmax and AUC values 44% higher than under fasting conditions. The extended halflife provides therapeutic concentrations for at least 72 h after administration, allowing less frequent dosing.
Distribution
Animal data suggest that it is well distributed in the body, with tissue concentrations exceeding the plasma concentration, except in bone, testes and brain. The ratio of intracellular:extracellular concentration in macrophages was estimated as 24:1.
Metabolism
The main metabolite is an antimicrobially active 25-desacetyl derivative. Although it induces liver cytochromes it is not an inducer of its own metabolism, which is mediated by an esterase. The peak concentration of 25-desacetyl rifapentine is about one-third of that of the unchanged drug, and is attained after about 11 h.
excretion
The main route of elimination is through the bile. In healthy volunteers about 70% of a 600 mg dose of 14C rifapentine was recovered in the feces, and less than 17% in the urine. There is evidence of enterohepatic recycling in humans.
Clinical Use
Tuberculosis (in combination with other antituberculosis drugs)부작용
Signs of teratogenic effects and fetal toxicity have been observed when administered during pregnancy to rats and rabbits. Rifapentine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.The most common adverse effect observed in combinations with other antimycobacterial agents was hyperuricemia, most probably due to pyrazinamide. Effects likely to be due to rifapentine were neutropenia (3.7% of patients) and hepatitis (increased transaminases in 1.6% of patients).
Rifapentine 준비 용품 및 원자재
원자재
준비 용품
Rifapentine 공급 업체
글로벌( 285)공급 업체
| 공급자 | 전화 | 이메일 | 국가 | 제품 수 | 이점 |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +8613288715578 |
sales@hbmojin.com | China | 12456 | 58 |
| Ouhuang Engineering Materials (Hubei) Co., Ltd | +8617702722807 |
admin@hbouhuang.com | China | 2880 | 58 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21691 | 55 |
| career henan chemical co | +86-0371-86658258 |
sales@coreychem.com | China | 29914 | 58 |
| Biochempartner | 0086-13720134139 |
candy@biochempartner.com | CHINA | 967 | 58 |
| Hebei Guanlang Biotechnology Co., Ltd. | +86-19930503282 |
alice@crovellbio.com | China | 8823 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-592-6051114 +8618959220845 |
sales@amoychem.com | China | 6387 | 58 |
| BOC Sciences | +1-631-485-4226 |
inquiry@bocsci.com | United States | 19553 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-61398051 +8613650506873 |
sales@chemdad.com | China | 39916 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49390 | 58 |
Rifapentine 관련 검색:
쿠레톡심메틸 아크릴산메틸 3차-아밀 메틸 에테르 메틸파라벤 벤설프론 메틸 리팜핀 티오판산-메틸 N-메틸피페라진 아세트산 메틸 메틸 파라싸이온(메틸 파라티온) 브롬화 메틸
Rifamycin Sodium
METHYL THIOPHENE-2-CARBOXYLATE
RIFAMYCIN SV
Methyl
RIFAPENTINE HYDROCHLORIDE,3-[[(4-CYCLOPENTYL-1-PIPERAZINYL)-IMINO]METHYL]RIFAMYCIN/RIFAPENTINE HYDROCHLORIDE,RIFAPENTINE
25-DESACETYL RIFAPENTINE
Rifapentine