|
|
| | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE Basic information |
| Product Name: | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE | | Synonyms: | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE;Cyclopentene-1,2-dicarboxylic acid anhydride;4,5,6,6a-Tetrahydro-1H-cyclopenta[c]furan-1,3(3aH)-dione;5,6-dihydro-4H-cyclopenta[c]furan-1,3-dione;5,6-dihydro-4H-cyclopenta[c]furan-1,3-quinone;NSC 291644;1-Cyclopentene-1,2-dicarboxylic anhydride 96%;1H,3H,4H,5H,6H-Cyclopenta[c]furan-1,3-dione | | CAS: | 3205-94-5 | | MF: | C7H6O3 | | MW: | 138.12 | | EINECS: | | | Product Categories: | Carbonyl Compounds;Carboxylic Acid Anhydrides;Organic Building Blocks;Building Blocks;Carbonyl Compounds;Carboxylic Acid Anhydrides;Chemical Synthesis;Organic Building Blocks | | Mol File: | 3205-94-5.mol | 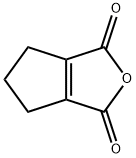 |
| | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE Chemical Properties |
| Melting point | 42-45 °C (lit.) | | Boiling point | 133-135 °C/10 mmHg (lit.) | | density | 1.2667 (rough estimate) | | refractive index | n20/D 1.4978(lit.) | | Fp | >230 °F | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | BRN | 123650 |
| | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE Usage And Synthesis |
| Uses | Valuable Diels-Alder dienophile, for example, in approach to the bicycloundecane system in taxanes. | | Purification Methods | If IR has OH peaks, then some hydrolysis to the diacid (m 178o) must have occurred. In this case reflux it with an appropriate volume of Ac2O for 30minutes, evaporate the Ac2O and distil it in vacuo. The distillate solidifies and can be recrystallised from EtOAc/hexane (1:1). The diacid distils without decomposition due to formation of the anhydride. The dimethyl ester has m 120-125o/11mm. [Askain Chem Ber 98 2322 1965, Beilstein 17/11 V 125.] |
| | 1-CYCLOPENTENE-1,2-DICARBOXYLIC ANHYDRIDE Preparation Products And Raw materials |
|