|
|
| | N-Cyclohexyl-4-methylbenzenesulfonamide Basic information |
| Product Name: | N-Cyclohexyl-4-methylbenzenesulfonamide | | Synonyms: | N-CYCLOHEXYL-P-TOLUENESULFONAMIDE;N-CYCLOHEXYL-P-TOLUENESULPHONAMIDE;n-cyclohexyltoluene-4-sulphonamide;N-Cyclohexyl-4-methylbenzenesulfonamide;TIMTEC-BB SBB006339;(tosylamino)cyclohexane;Benzenesulfonamide, N-cyclohexyl-4-methyl-;n-cyclohexyl-4-methyl-benzenesulfonamid | | CAS: | 80-30-8 | | MF: | C13H19NO2S | | MW: | 253.36 | | EINECS: | 201-268-3 | | Product Categories: | | | Mol File: | 80-30-8.mol | 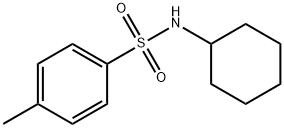 |
| | N-Cyclohexyl-4-methylbenzenesulfonamide Chemical Properties |
| Safety Statements | 22-24/25 | | RTECS | XT5617000 | | TSCA | Yes | | HS Code | 2935.90.9500 |
| | N-Cyclohexyl-4-methylbenzenesulfonamide Usage And Synthesis |
| Chemical Properties | white or yellowish solid | | Synthesis Reference(s) | The Journal of Organic Chemistry, 46, p. 342, 1981 DOI: 10.1021/jo00315a024
Synthesis, p. 481, 1993 | | General Description | Yellowish-brown fused mass or white crystalline solid. Little or no odor. | | Air & Water Reactions | Insoluble in water. | | Reactivity Profile | Organic amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). | | Fire Hazard | Flash point data for N-Cyclohexyl-4-methylbenzenesulfonamide is not available; however, N-Cyclohexyl-4-methylbenzenesulfonamide is probably combustible. |
| | N-Cyclohexyl-4-methylbenzenesulfonamide Preparation Products And Raw materials |
|