|
|
| | calcium thiosulphate Basic information |
| Product Name: | calcium thiosulphate | | Synonyms: | CalciuM thiosulfate, pure, 30-50% solution in water;calcium thiosulphate;Thiosulfuric acid (H2S2O3), calcium salt (1:1);Calcium thiosulfate hexahydrate.;calcium hyposulfite;calcium thiosulfate [ca(s2o3)];tecesal;thiosulfuric acid, calcium salt | | CAS: | 10124-41-1 | | MF: | CaH4O3S2 | | MW: | 156.23 | | EINECS: | 233-333-7 | | Product Categories: | | | Mol File: | 10124-41-1.mol | 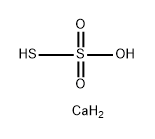 |
| | calcium thiosulphate Chemical Properties |
| Melting point | decomposes [CRC10] | | density | 1.870 | | solubility | soluble in H2O; insoluble in ethanol | | form | triclinic crystals | | color | triclinic crystals, crystalline | | Water Solubility | 100g/100mL H2O (3°C), decomposed by hot water; insoluble alcohol [MER06] [CRC10] | | EPA Substance Registry System | Calcium thiosulfate (10124-41-1) |
| | calcium thiosulphate Usage And Synthesis |
| Chemical Properties | tricl crystal(s); when dry, decomposes on standing to form a yellow crust on the surface; spontaneously decomposed at 43°C–49°C; used to treat dermatitis and jaundice caused by arsphenamine [MER06] | | Purification Methods | Recrystallise it from water below 60o in a N2 atmosphere, followed by drying with EtOH and Et2O. Store it in a refrigerator. The hexahydrate can decompose spontaneously at 43-49o. Store it in a cool closed container. [Pethybridge & Taba J Chem Soc, Faraday Trans 1 78 1331 1982.] |
| | calcium thiosulphate Preparation Products And Raw materials |
|