|
|
| | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane Basic information |
| Product Name: | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane | | Synonyms: | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane;3,3,6,6,9,9-Hexamethyl-1,2,4,5-tetraoxacyclononane;P-1230;1,2,4,5-Tetroxonane, 3,3,6,6,9,9-hexamethyl-;3,3,6,6,9.9-Hexamethyl-1,2,4,5-tetraoxocyclononane;Einecs 244-954-8;3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane(purity is 52%~100%);3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane[Content (< 52%) and type A diluent (> 48%)] | | CAS: | 22397-33-7 | | MF: | C11H22O4 | | MW: | 218.29 | | EINECS: | 244-954-8 | | Product Categories: | | | Mol File: | 22397-33-7.mol | 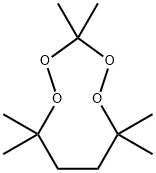 |
| | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane Chemical Properties |
| | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane Usage And Synthesis |
| Reactivity Profile | Ketones, such as 3,3,6,6,9,9-HEXAMETHYL-1,2,4,5-TETRAOXACYCLONONANE, are reactive with many acids and bases liberating heat and flammable gases (e.g., H2). The amount of heat may be sufficient to start a fire in the unreacted portion of the ketone. Ketones react with reducing agents such as hydrides, alkali metals, and nitrides to produce flammable gas (H2) and heat. Ketones are incompatible with isocyanates, aldehydes, cyanides, peroxides, and anhydrides. They react violently with aldehydes, HNO3, HNO3 + H2O2, and HClO4. |
| | 3,3,6,6,9,9-hexamethyl-1,2,4,5-tetroxonane Preparation Products And Raw materials |
|