|
|
| | NEOEVONINE Basic information |
| Product Name: | NEOEVONINE | | Synonyms: | NEOEVONINE;6-O-Deacetylevonine;Evorine;Neoevonin;8,11-Epoxy-9,12-ethano-11,15-methano-11H-[1,8]dioxacycloheptadecino[4,3-b]pyridine-5,17,22-trione, 13,14,21-tris(acetyloxy)-12-[(acetyloxy)methyl]-7,8,9,10,12,13,14,15,18,19-decahydro-10,20-dihydroxy-8,18,19,20-tetramethyl-, (8R,9R,10R,11S,12S,13R,14R,15S,18S,19S,20S,21S)- (9CI) | | CAS: | 33540-08-8 | | MF: | C34H41NO16 | | MW: | 719.69 | | EINECS: | | | Product Categories: | | | Mol File: | 33540-08-8.mol | 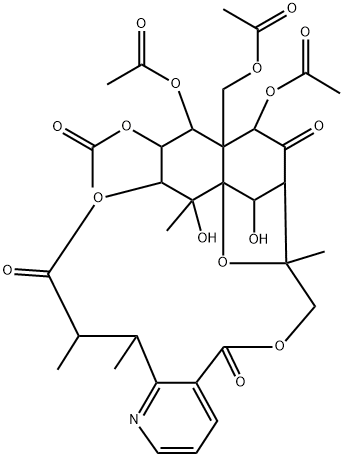 |
| | NEOEVONINE Chemical Properties |
| Melting point | 264-SoC | | Boiling point | 800.1±65.0 °C(Predicted) | | density | 1.44±0.1 g/cm3(Predicted) | | pka | 11.97±0.70(Predicted) |
| | NEOEVONINE Usage And Synthesis |
| Description | A second macrocyclic alkaloid found in Euonymus sieboldiana Blume, this base
also crystallizes as colourless crystals and is best purified by recrystallization
from EtOH. It is dextrorotatory having [α]D + 24.9° (c 1.27, CHCI3). The
ultraviolet spectrum is very similar to that of the preceding alkaloid with absorp_x0002_tion maxima at 224 and 26S mJi. The structure, as determined from the NMR
and mass spectra is virtually identical with that of neoeuonymine, the only
difference being the replacement of an acetoxy group with a ketone group in the
reduced naphthalene moiety. | | References | Sugiura et al., Tetrahedron Lett., 2733 (1971)
Stereochemistry:
Wada et al., Tetrahedron Lett., 3131 (1971) |
| | NEOEVONINE Preparation Products And Raw materials |
|