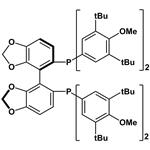
- (R)-DTBM-Segphos
-
- $0.00 / 1kg
-
2023-12-07
- CAS:566940-03-2
- Min. Order: 1kg
- Purity: 98%, ee>99
- Supply Ability: 200kg
|
| | (R)-DTBM-SEGPHOS Basic information | | Reaction |
| Product Name: | (R)-DTBM-SEGPHOS | | Synonyms: | (R)-(-)-5,5'-Bis[di(3,5-di-t-butyl-4-methoxyphenyl)phosphino]-4,4'-bi-1,3-benzodioxole,min.98%(R)-DTBM-SEGPHOS;(R)-DTBM-SEGPHOS(R);[(4R)-(4,4μ-bi-1,3-benzodioxole)-5,5μ-diyl]bis[bis(3,5-di-tert-butyl-4-methoxyphenyl)phosphine], (R)-(-)-5,5μ-Bis[di(3,5-di-tert-butyl-4-methoxyphenyl)phosphino]-4,4μ-bi-1,3-benzodioxole;(R)-(-)-5,5'-Bis[di(3,5-di-t-butyl-4-Methoxyphenyl)phosphino]-4,4'-bi-1,3-benzodioxole;(4R)-[4,4'-Bi-1,3-benzodioxole]-5,5'-diylbis[bis[3,5-bis(1,1-dimethylethyl)-4-methoxyphenyl]phosphine;(R)-()-5,5′-Bis[di(3,5-di-tert-butyl-4-methoxyphenyl)phosphino]-4,4′-bi-1,3-benzodioxole;(R)-DTBM-SEGPHOS;[(4R)-(4,4′-bi-1,3-benzodioxole)-5,5′-diyl]bis[bis(3,5-di-tert-butyl-4-methoxyphenyl)phosphine] | | CAS: | 566940-03-2 | | MF: | C74H100O8P2 | | MW: | 1179.55 | | EINECS: | | | Product Categories: | Segphos Series;Chiral Phosphine | | Mol File: | 566940-03-2.mol | 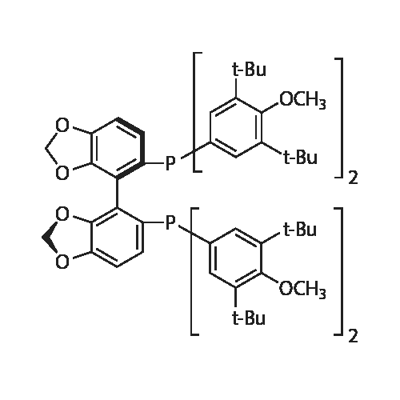 |
| | (R)-DTBM-SEGPHOS Chemical Properties |
| Melting point | 126-128°C | | Boiling point | 987.3±65.0 °C(Predicted) | | form | Powder | | color | off-white |
| WGK Germany | 3 | | HS Code | 2932.99.7000 |
| | (R)-DTBM-SEGPHOS Usage And Synthesis |
| Reaction |
- Rhodium catalyzed chemo-, regio, and entantioselective [2 + 2 + 2] cycloaddition of alkynes with isocyanates.
- With copper, enantioselective cross Aldol-type reaction of acetonitrile.
- With copper, enantioselective vinylsilane alkenylation of aldehydes.
- Gold carbene mediated stereoselective cyclopropanation of propargyl esters.
- With copper, enantioselective 1,2-reduction of ketones, and 1,4-reduction of a α,β-usaturated esters.
- With copper, catalytic enantioselective Mannich-type reaction.
- Enantioselective fluorination of b β-keto esters, tert-butoxycarbonyl lactones and lactmes with Sodeoka's Pd-aqua complex and a fluorinating reagent.
- Rh-catalyzed intramolecular olefin or carbonyl hydroacylation.
- Pd-catalyzed γ-arylation of β,γ-unsaturated ketones.
- Involved in numerous conjugate alkynylation, and ring-opening alkynylation of azabenzonorbornadienes.
- Involved in asymmetric hydroamination of bicyclic alkenes/dienes,13a diamination of conjugated dienes,13b and hydroalkoxylation/hydrosulfenylation of allenes.
The actions in the following figures are corresponding to the above ones in sequence.
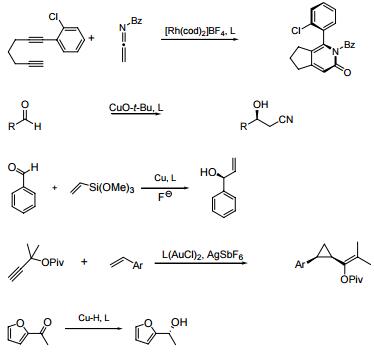
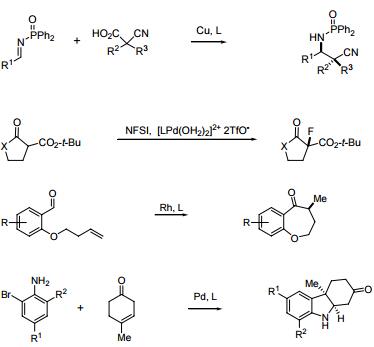
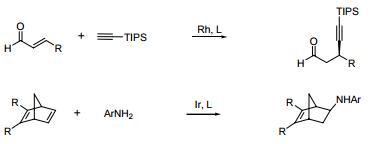
| | Uses | Catalyst involved in:
- [3,3]-Sigmatropic rearrangements using cyclopropane probes
- Asymmetric intramolecular hydroacylation of ketoaldehydes
Reactant involved in:
- The synthesis of gold-diphosphine complexes for use as catalysts
- Cycloaddition of allenenes to yield alkylidenecyclobutanes
|
| | (R)-DTBM-SEGPHOS Preparation Products And Raw materials |
|