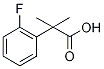
2-(2-Fluorophenyl)-2-methylpropionicacid synthesis
- Product Name:2-(2-Fluorophenyl)-2-methylpropionicacid
- CAS Number:870849-49-3
- Molecular formula:C10H11FO2
- Molecular Weight:182.19
861901-19-1
19 suppliers
$90.00/100mg

870849-49-3
34 suppliers
$50.00/100mg
Yield:870849-49-3 5.799 g
Reaction Conditions:
with water;sodium hydroxide in 1,4-dioxane; for 24 h;
Steps:
1 Dimethylation and hydrolysis
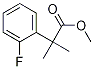
A solution of methyl 2-(2- fluorophenyl)acetate (6.373 g, 37.89 mmol) and methyl iodide (5.90 mL, 94.8 mmol, 2.5 equiv) in anhydrous THF (25 mL) was treated portionwise with sodium hydride (60% suspension in mineral oil, 3.32 g, 83.0 mmol, 2.2 equiv) at 0 °C, warmed to room temperature and reacted for 24 h. The reaction mixture was transferred into a separatory funnel with ice/water and acidified with 1.0 M HC1 (30 mL). The product was extracted with ethyl acetate (3 x 30 mL). The combined organic layers were washed brine, filtered through cotton, and concentrated under reduced pressure to yield light yellow oil. The oil was dissolved in 16 mL of l,4-dioxane and 19.0 mL of 6.0 M NaOH (113.7 mmol, 3.0 equiv) was added. The mixture was heated at 95 °C for 24 h. 30 mL of 3.0 M HC1 was added and the mixture was extracted with dichloromethane (3 x 20 mL). Combined organic fractions were washed with brine (30 mL), filtered through cotton and concentrated under reduced pressure to yield a crude mixture which was purified by flash chromatography (hexanes/ethyl acetate), 5.799 g of 2-methyl-2-(2-fluorophenyl)propanoic acid (31.83 mmol, 84% over two steps).
References:
WO2019/99760,2019,A1 Location in patent:Paragraph 0068; 0070
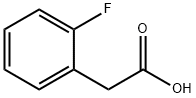
451-82-1
341 suppliers
$6.00/5g

74-88-4
341 suppliers
$15.00/10g

870849-49-3
34 suppliers
$50.00/100mg