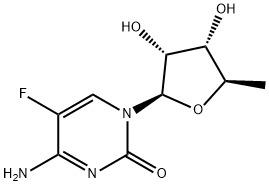
5'-Deoxy-5-fluorocytidine synthesis
- Product Name:5'-Deoxy-5-fluorocytidine
- CAS Number:66335-38-4
- Molecular formula:C9H12FN3O4
- Molecular Weight:245.21
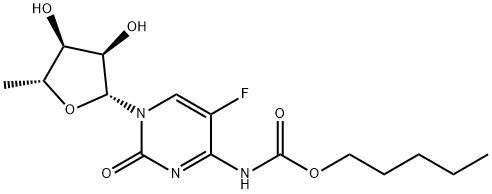
154361-50-9
788 suppliers
$5.00/1g

66335-38-4
350 suppliers
$8.00/250mg
Yield:66335-38-4 90%
Reaction Conditions:
with potassium carbonate in methanol;N,N-dimethyl-formamide for 24 h;Heating / reflux;
Steps:
9
Capecitabine (5'-deoxy-5-fluoro-N4-(pentyloxycarbonyl)cytidine, 4.5 g, 12.5 mmol) was suspended in methanol (250 mL) and DMF (10 mL) together with potassium carbonate (8.8 g, 63.77 mmol) and the solution heated under reflux for 24 h. The solution was then cooled and evaporated to dryness (below 45 °C). The residue was redissolved in hot methanol, filtered and washed with hot methanol. The filtrate was preabsorbed onto silica gel and puified by flash chromatography, eluting with 50 % methanol / ethyl acetate to give 3.0 g (90 %) of l-[3,4-dihydroxy-5-methyl- tetrahydrofuran-2-yl]-4-amino-lH-pyrimidin-2-one. This material was suspended in chloroform (125 mL) and the solution heated to 50 0C. Acetic acid (2 mL, 34 mmol) was added and after 10 minutes at 50 0C, acetyl chloride (20 mL, 206 mmol) was added. The suspension was stirred at 50 0C for 7 h and then at 20 °C for 72 h. Ether (100 mL) was added and the solid filtered and washed with ether to give 4.0 g (90 %) l-[3,4-Diacetoxy-5-methyl-tetrahydrofuran-2-yl]-4-amino-lH-pyrimidin-2-one hydrochloride. This material (2.07g, 5.7 mmol) together with 5-nitrothien-2- ylmethanol (1.47 g, 9.3 mmol), was dissolved in pyridine (1.4 mL, 17.4 mmol) and DCM (15 mL). Phosgene solution (3.6 mL of a 2M solution in toluene, 7.2 mmol) was slowly added to the above cooled (O0C) solution, the solution was stirred at O0C for 2.5 h and then a further 3.6 mL of phosgene solution added and the solution stirred at O0C for a further 2h and refrigerated for 18 h. The solution was partitioned (ethyl EPO
References:
ANGIOGENE PHARMACEUTICALS LIMITED;THE GRAY LABORATORY CANCER RESEARCH TRUST WO2006/32921, 2006, A1 Location in patent:Page/Page column 37-38
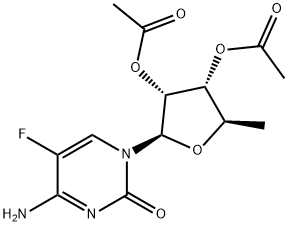
161599-46-8
442 suppliers
$6.00/250mg

66335-38-4
350 suppliers
$8.00/250mg
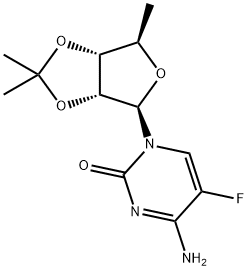
66335-37-3
44 suppliers
inquiry

66335-38-4
350 suppliers
$8.00/250mg