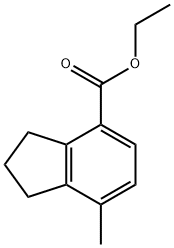
2,3-Dihydro-7-methyl-1H-indene-4-carboxylic acid ethyl ester synthesis
- Product Name:2,3-Dihydro-7-methyl-1H-indene-4-carboxylic acid ethyl ester
- CAS Number:71042-72-3
- Molecular formula:C13H16O2
- Molecular Weight:204.26
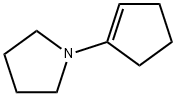
7148-07-4
85 suppliers
$57.79/10g
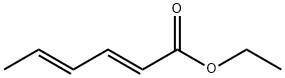
2396-84-1
154 suppliers
$11.00/10g

71042-72-3
39 suppliers
$45.00/50mg
Yield:71042-72-3 58%
Reaction Conditions:
Stage #1: 1-pyrrolidinocyclopent-1-ene;(2E,4E)-ethyl hexa-2,4-dienoate in 5,5-dimethyl-1,3-cyclohexadiene;Reflux;
Stage #2: with sulfur at 250; for 2 h;
Steps:
1.1 Step 1. Synthesis of ethyl 7-methyl-2,3-dihydro-1H-indene-4-carboxylate (1-1)
A mixture of 13 ethyl sorbate (25.0 mL, 170 mmol, TCI reagent) in 14 xylene (100 mL) and 15 1-pyrrolidino-1-cyclopentene (24.8 mL, 170 mmol, TCI agent) was stirred at reflux overnight. After a reaction was completed, a volatile solvent was evaporated under reduced pressure. 16 EtOAc was added into a resulting mixture. An organic layer was washed with brine, after which a resulting product was dried over anhydrous MgSO4, filtered and concentrated under vacuum. A crude compound was used in a following step without an additional purification. S8 (5.45 g, 170 mmol) was added into the crude compound. A reaction mixture was stirred at 250 °C for 2 hours. After a reaction was completed, the resulting mixture was distilled under reduced pressure, so as to obtain the 17 title compound (1-1) (20.0 g, 97.9 mmol, 58%). (0078) 1H NMR (400 MHz, CDCl3); δ 7.76 (d, J = 7.6 Hz, 1H), 7.03 (d, J = 8.0 Hz, 1H), 4.34 (q, J = 7.2 Hz, 2H), 3.30 (t, J = 7.6 Hz, 2H), 2.84 (t, J = 7.6 Hz, 2H), 2.30 (s, 3H), 2.12-2.05 (m, 2H), 1.38 (t, J = 7.2 Hz, 3H)
References:
EP3404033,2018,A1 Location in patent:Paragraph 0077-0078

7148-07-4
85 suppliers
$57.79/10g

2396-84-1
154 suppliers
$11.00/10g

71042-72-3
39 suppliers
$45.00/50mg
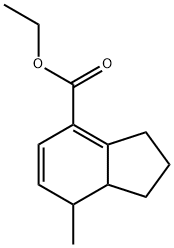
71042-73-4
0 suppliers
inquiry