Ethyl propiolate derivatisation for the analysis of varietal thiols in wine
Nov 12,2019
1. Introduction
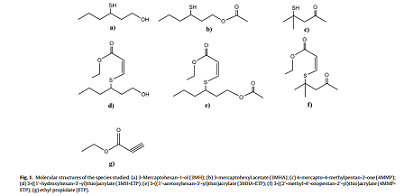
Amongst all the different aroma compound classes present in wine, varietal thiols [e.g. 3-mercaptohexan-1-ol (3MH), 3- mercaptohexyl acetate (3MHA) and 4-mercapto-4-methyl pentan- 2-one (4MMP)] are undoubtedly one ofthe most widely studied [1]. Varietal thiols (Fig. 1) are present in grape juice as non-volatile Sglutathionylated, S-cysteinylated and recently identified S-glycine cysteinylated conjugated precursors [2–5] and released during fermentation via yeast action [6,7]. These molecules elicit a vast interest in the wine science community for their extremely low sensory thresholds and positive sensory impact.
The analytical quantification of 3MH, 3MHA and 4MMP has always posed the most significant obstacle for their study: their level in wine is often extremely low [1] and the presence of the thiol group makes them fairly susceptible to oxidation [8]. Many efforts have been put into this task with all the different tools currently available to analytical chemists being applied [9–13]. As the presence of the free thiol group represents the most important issue for the analytical quantification several derivatising agents have been tested. The most effective methods apply organo-mercury compounds (p-hydroxymercuribenzoate – pHMB and p-aminophenylmercuric acetate): these are, to the best of our knowledge, the only two methods capable of detecting all the three varietal thiols at levels sufficiently low to be applicable in wine matrices [14,15]. However, the use of mercury constitutes a health and safety risk in the laboratory and additionally these methods are very time-consuming.
Another very promising derivatising agent suggested in the literature is the 2,3,4,5,6-pentafluorobenzyl bromide. This molecule has been used in on-fibre, on-cartridge and liquid/liquid based methods [9–11]. Nevertheless all these methods failed in achieving the quantification of all three thiols or were extremely time consuming, leaving the mercury based methods the only way to quantify them.
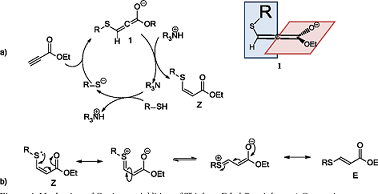
Ethyl propiolate (ETP) is a compound largely used in organic chemistry and it has been recently applied as a derivatising agent for the thiol group [16–19]. Very recently a new propiolate-based derivatising agent (2-naphthyl propiolate) was also developed to quantify captopril in human plasma via HPLC–UV detection [20]. The reaction mechanism relies on the nucleophilic attack of the thiolate group on to the -carbon of the propiolate generating two stable alkylthioacrylate species ((Z) and (E)), with the (Z) speciesbeing the favoured one [21,22]. The kinetics are controlled by the thiol group dissociation constant. Generally, ETP reacts under basic pH (reaction pH > pKa of the thiol). The main advantages connected with this reaction are its rapidity, effectiveness and the generation of UV active species [23].
To the best of our knowledge this method has only been applied for the derivatisation of thiol species, other than the ones we are currently investigating, and followed by detection via LC–UV. However, when this approach was preliminarily tried, the detection limit reached without pre-concentration was not satisfactory and when a preliminary SPE step was introduced the presence of other species absorbing at the same wavelength made the quantification of the derivatised thiols impossible. The intrinsic low molecular weight of ETP led us to consider the possibility of detecting small thiols derivatised with ETP via gas chromatography. This hypothesis was never reported before.
In this paper, the derivatisation of the important varietal thiols with ETP followed by GC–MS concurrent stable isotope dilution quantification is reported for the first time. The effect of the pH on the derivatisation reaction, on the potential hydrolysis of 3MHA and the nature of the cartridge adopted were investigated. Finally a stable isotope dilution SPE-GC–MS assay able to quantify the three important thiols was developed, optimised and validated.
2. Experimental
The sulfur compounds studied 3-mercaptohexan-1-ol(3MH), 3- mercaptohexyl acetate (3MHA) and 4-mercapto-4-methyl pentan- 2-one were purchased from Acros Organics (Geel, Belgium), Oxford Chemical(Hartlepool, UK), andAldrich (Castle Hill, NSW,Australia), respectively. The internal standards d2-3-mercaptohexan-1-ol (d2-3MH), d2-3-mercaptohexyl acetate ([d2-3MHA) and d10-4- mercapto-4-methyl pentan-2-one (d10-4MMP) were synthesised at The University of Auckland [24,25], while 4-methoxy-2- methyl-2-mercaptobutane (4M2M2MB) was kindly donated by Dr Tominaga. 5,5 -Dithio-bis-(2-nitrobenzoic acid) (99%) was purchased from Acros Organics (Geel, Belgium). Hydrochloric acid (37%, reagent grade), sodium hydroxide (pellets, ≥99%, reagent grade), sodium acetate trihydrate (99.5–100.5%) and sodium sulfate anhydrous (powder, extra pure, 98.5–100.5%) were obtained from Scharlau (Barcelona, Spain). Ethyl propiolate (ETP) and butylated hydroxyanisole (BHA) were purchased from Aldrich (Castle Hill, NSW, Australia). 4-Hydroxymercuribenzoic acid sodium salt (≥95.0% Hg) came from Sigma (Castle Hill, NSW, Australia), while DOWEX® (1 × 2, Cl−-form, strongly basic, 50–100 mesh) came from Acros Organics (Thermo Fisher Scientific, Auckland, New Zealand). Methanol (HPLC grade, Scharlau, Barcelona, Spain), ethyl acetate (≥99.7%, LC–MS CHROMASOLV®, Fluka, Castle Hill, NSW, Australia) and dichloromethane (for gas chromatography, SupraSolv®, Merck, Darmstadt, Germany) were used as solvents. TRIS (ultrapure, ≥99.9%) and l-cysteine BioChemica was provided by Applichem (Darmstadt, Germany) The SPE cartridges tested were SupelcleanTM ENVI-18 (6 mL cartridge volume; 1 g sorbent; Supelco, Castle Hill, NSW, Australia), LiChrolut EN (40–120 m; 6 mL cartridge volume; 500 mg sorbent; Merck, Darmstadt, Germany), Strata SDB-L Styrene-Divinylbenzene Polymer (100 m, 260A; 6 mL cartridge volume; 500 mg sorbent; Phenomenex, Torrance, CA, USA) as well as a self-packed C18 (6 mL cartridge volume; 1 g sorbent, Zeochem, Uetikon, Switzerland).
Nitrogen (food grade) and helium (instrument grade) were sourced from BOC Gases New Zealand Ltd. (Auckland, New Zealand).
Solutions were prepared using Grade 1 water (BARNSTEAD® NANOpure DIamondTM Water Purification System, Thermo Scientific, USA) with a resistivity of 18.2 M/cm at 25 ◦C or absolute ethanol (≥99.5%, Univar, Ajax Finechem, Auckland, New Zealand). Model white wine base was prepared by dissolving 5 g/L of l-(+)- tartaric acid (≥99%, Scharlau, Barcelona, Spain) in an ultrapure water/ethanol (88:12 v/v) solution and adjusted to pH 3.2, using NaOH (10 N and 1 N) and HCl (1 N).
3. Results and discussion
The reaction between ETP and varietal thiols meets some important analytical requirement such as selectivity and rapidity; nonetheless it poses some serious issues mainly connected with the basic pH required for the reaction to occur.
In order to select the best SPE cartridge to be used to reach the sensitivity needed, a working pH of 9 was chosen. Four cartridges were tested: SupelcleanTM ENVI-18 1 g/6 mL, LiChrolut EN (40–120 m) 500 mg/6 mL, Strata SDB-L Styrene–divinylbenzene polymer (100 m, 260A) 500 mg/6 mL as well as one self-packed C18 1 g/6 mL. Two replicated measurements of a wine spiked with 400 ng/L of three thiols were performed for each cartridge.
Cartridges Lichrolut EN and Strata SDB-L were excluded as they produced wider peaks and a higher number of peaks, respectively, that made the identification and quantification of the target analytes more difficult. ENVI-18 cartridge was chosen to optimise and validate this method.
The second criterion that was then evaluated was the pH of the derivatisation. It has been reported that the yield of this reaction is strongly governed by the availability of free ETP in the media and by the presence of the thiolate group acting as nucleophile [16]. Four different pH values (i.e. 7, 8, 9, 10) were tested in a wine sample spiked with 400 ng/L of the three thiols in order to verify the best derivatisation conditions. Three replicated measurements were performed and Fig. 2 depicts the evolution profile ofthe derivatisation reaction for the three analytes. The different profile shown by 3MH and 4MMP could be related to hydrogen bonding between the thiol group and the carbonyl 4MMP is able to make, which could potentially cause a minor influence of the pH on this species.
Finally, since a high pH seemed to be required to ensure a good derivatisation yield, the effect of such a pH on the stability of the 3MHA-ETP adduct against ester hydrolysis had to be taken into consideration. Three different pH values were taken into account (i.e. pH 8, 9 and 10) and the evolution of 3MHA-ETP over the internal standard (i.e. d2-3MH-ETP) was followed over four time points (i.e. 0, 15, 30 and 45 min waiting time after 10 min stirring) in a model wine matrix spiked with 400 ng/L of 3MHA. The time points were chosen according to an estimation of the maximum time between the pH modification, when the hydrolysis could start, and the percolation of the derivatised thiols in the organic phase, when no further hydrolysis should occur. Each time point was replicated two times. Fig. 3 shows the evolution profile of 3MHA-ETP at pH 10 against d2-3MH-ETP. Additionally, no 3MH coming from the hydrolysis of 3MHA was identified in any of the samples. Since no hydrolytic effect was observed for 3MHA at pH 10, and this pH was ensuring the highest response for all the three thiols, an operating pH of 10 was chosen for the method validation.
4. Conclusion
Thepresentpaper reports for thefirsttime theuse of ethylpropiolate as a derivatising agentfor varietalthiols in grape products. The use of gas chromatographic approaches to follow the derivatised thiols here described was also never reported in the literature.
The method developed is rapid and simple and has performances that make it perfectly suitable for the analysis of oenological samples. Derivatisation of 4MMP proved to be particularly challenging with the degree of derivatisation (and therefore sensitivity) dependent upon the wine matrix being analysed. Further studies are required in order to optimise this reaction for 4MMP.
Finally, the new method is much safer for the analyst as it does not require mercury derived species, rendering it perfectly applicable in the wider wine-related environments such as wineries and analytical service laboratories.
References
[1] A. Roland, R. Schneider, A. Razungles, F. Cavelier, Chem. Rev. 111 (2011) 7355.
[2] T. Tominaga, C. Peyrot des Gachons, D. Dubourdieu, J. Agric. Food Chem. 46 (1998) 5215.
[3] C. Peyrot des Gachons, T. Tominaga, D. Dubourdieu, J. Agric. Food Chem. 50 (2002) 4076.
[4] B. Fedrizzi, K.H. Pardon, M.A. Sefton, G.M. Elsey, D.W. Jeffery, J. Agric. Food Chem. 57 (2009) 991.
[5] D.L. Capone, K.H. Pardon, A.G. Cordente, D.W. Jeffery, J. Agric. Food Chem. 59 (2011) 11204.
[6] G. Winter, T. van der Westhuizen, V.J. Higgins, C. Curtin, M. Ugliano, Aust. J. Grape Wine Res. 17 (2011) 285.
[7] M. Roncoroni, M. Santiago, D.O. Hooks, S. Moroney, M.J. Harsch, S.A. Lee, K.D. Richards, L. Nicolau, R.C. Gardner, Food Microbiol. 28 (2011) 926.
[8] M. Nikolantonaki, A.L. Waterhouse, J. Agric. Food Chem. 60 (2012) 8484.
[9] L. Mateo-Vivaracho, V. Ferreira, J. Cacho, J. Chromatogr. A 1121 (2006) 1.
[10] L. Mateo-Vivaracho, J. Cacho, V. Ferreira, J. Chromatogr. A 1146 (2007) 242.
[11] L. Mateo-Vivaracho, J. Cacho, V. Ferreira, J. Chromatogr. A 1185 (2008) 9.
[12] B. Fedrizzi, G. Versini, I. Lavagnini, G. Nicolini, F. Magno, Anal. Chim. Acta 596 (2007) 291.
[13] B. Fedrizzi, G. Versini, I. Lavagnini, D. Badocco, G. Nicolini, F. Magno, Anal. Chim. Acta 621 (2008) 38.
[14] T. Tominaga, M.L. Murat, D. Dubourdieu, J. Agric. Food Chem. 46 (1998) 1044.
[15] R. Schneider, Y. Kotseridis, J.L. Ray, C. Augier, R. Baumes, J. Agric. Food Chem. 51 (2003) 3243.
[16] C.K. Zacharis, P.D. Tzanavaras, D.G. Themelis, J. Pharm. Biomed. Anal. 50 (2009) 384.
[17] P.D. Tzanavaras,A. Tsiomlektsis, C.K. Zacharis, J. Pharm. Biomed.Anal. 53 (2010) 790.
[18] P.D. Tzanavaras, C.K. Zacharis, D.G. Themelis, Anal. Lett. 43 (2010) 1889.
[19] C.K. Zacharis, P.D. Tzanavaras, A. Zotou, Anal. Chim. Acta 690 (2011) 122.
[20] N. Rastkari, M. Khoobi, A. Shafiee, M.R. Khoshayand, R. Ahmadkhaniha, J. Chromatogr. B 932 (2013) 144.
[21] W.E. Truce, G.J.W. Tichenor, J. Org. Chem. 37 (1972) 2391.
[22] P.D. Halphen, T.C. Owen, J. Org. Chem. 38 (1973) 3507.
[23] T.C. Owen, Bioorg. Chem. 36 (2008) 156.
[24] Y. Kotseridis, J.L. Ray, C. Augier, R.L. Baumes, J. Agric. Food Chem. 48 (2000) 5819.
[25] K.R. Hebditch, L. Nicolau, M.A. Brimble, J. Labelled Compd. Radiopharm. 50 (2007) 237.
[26] M. Herbst-Johnstone, L. Nicolau, P.A. Kilmartin, Am. J. Enol. Vitic 62 (2011) 495.
[27] A. Hubaux, G. Vos, Anal. Chem. 42 (1970) 849.
[28] I. Lavagnini, B. Fedrizzi, G. Versini, F. Magno, Rapid Commun. Mass Spectrom. 23 (2009) 1167.
[29] S. Jouanneau, R.J. Weaver, L. Nicolau, M. Herbst-Johnstone, F. Benkwitz, P.A. Kilmartin, Aust. J. Grape Wine Res. 18 (2012) 329.
[30] F. Benkwitz, T. Tominaga, P.A. Kilmartin, C. Lund, M. Wohlers, L. Nicolau, Am. J. Enol. Vitic 63 (2012) 62.
[31] T. Allen, M. Herbst-Johnstone, M. Girault, P. Butler, G. Logan, S. Jouanneau, L. Nicolau, P.A. Kilmartin, J. Agric. Food Chem. 59 (2011) 10641.
[32] Office International de la Vigne et du Vin, Recueil des methodes internationals d’analyse des vins et des mouts, MA-F-AS1-06-PROVAL, 2003.
- Related articles
- Related Qustion
- Ethyl propiolate: A Versatile Compound with Applications in Pyrazole Synthesis and Gasoline Additives Feb 5, 2024
Ethyl propiolate's synthesis involves esterification or transesterification reactions. It is utilized in pyrazole synthesis and as a gasoline additive for improved combustion performance.
- Ethyl propiolate: properties, applications as analytical reagent and safety Oct 8, 2023
Ethyl propiolate is a versatile derivatizing agent for thiols, used in chemical and biological research, as well as an analytical reagent for thiol determination.
Phenylboronic acid or benzeneboronic acid, abbreviated as PhB(OH)2 where Ph is the phenyl group C6H5-, is a boronic acid containing a phenyl substituent and two hydroxyl groups attached to boron.....
Nov 12,2019Chemical ReagentsLinalool is a component of many essential oils, including orange, lavender, rose, rosewood, and coriander. The main problem the Cosmetics Database has with linalool is the risk of skin irritation and allergic reactions.....
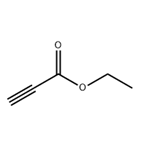
Nov 12,2019Organic ChemistryEthyl propiolate
623-47-2You may like
Ethyl propiolate manufacturers
- Ethyl propiolate
-

- $0.00 / 25KG
- 2025-06-20
- CAS:623-47-2
- Min. Order: 1KG
- Purity: 98%
- Supply Ability: 5MT/Month
- Ethyl propiolate
-
- $0.00 / 1kg
- 2025-04-04
- CAS:623-47-2
- Min. Order: 1kg
- Purity: 98%
- Supply Ability: 1Ton
- Ethyl propiolate
-

- $15.00 / 1KG
- 2021-07-13
- CAS:623-47-2
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton