1135-66-6
中文名称
异长叶烯
英文名称
Isolongifolene
CAS
1135-66-6
EINECS 编号
214-494-2
分子式
C15H24
MDL 编号
MFCD00042616
分子量
204.35
MOL 文件
1135-66-6.mol
更新日期
2026/05/06 11:35:55
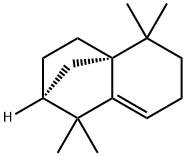 1135-66-6 结构式
1135-66-6 结构式
基本信息
中文别名
(2S)-1,3,4,5,6,7-六氢-1,1,5,5-四甲基-2H-2,4A-亚甲基萘异长叶烯
(-)-异长叶烯
英文别名
(1R)-2,2,7,7-TETRAMETHYLTRICYCLO[6.2.1.0(1.6)]UNDEC-5-ENE(-)-ISOLONGIFOLENE
ISOLONGIFOLENE
(-)-Isolongifoline
(2s)-1,3,4,5,6,7-hexahydro-1,1,5,5
(2s)-1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-2h-2,4a-methanonaphthalene
2H-2,4a-Methanonaphthalene, 1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-
2H-2,4a-Methanonaphthalene, 1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-, (2S,4aR)-(-)-
2H-2,4a-Methanonaphthalene, 1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-, (2S-cis)-
3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-4a-methanonaphthalen(2s)-2h-1
4a-Methanonaphthalene,1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-,(2S)-2H-2
Isolongipholene
C15 H24, 2H-2,4a-Methanonaphthalene, 1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-, (2S)-
ISOLONGIFOLENE(SG)
(2s)-2h-1,3,4,5,6,7-hexahydro-1,1,5,5-tetramethyl-4a-methanonaphthalen
1,3,4,5,6,7-Hexahydro-1,1,5,5-tetramethyl-2H-2,4a-methanonaphthalene
ISOLONGIFOLENE WITH GC
(2S,4aR)-1,2,3,4,4a,5,6,7-Octahydro-1,1,5,5-tetramethyl-2,4a-methanonaphthalene
(2S,4aR)-1,3,4,5,6,7-Hexahydro-1,1,5,5-tetramethyl-2H-2,4a-methanonaphthalene
(2S,4aR)-1,3,4,5,6,7-Hexahydro-1,1,5,5-tetramethyl-2H-2α,4aα-methanonaphthalene
所属类别
香精与香料:萜烯烃物理化学性质
外观性质微黄色液体,不溶于水,溶于乙醇等有机溶剂。沸点:113~114℃折光率:1.499密度:0.930闪点:49 °C
沸点255-256 °C
密度0.930 g/mL at 20 °C(lit.)
蒸气压5.49Pa at 25℃
折射率n20/D 1.499
闪点49°C
储存条件2-8°C
溶解度DMSO: 10 mg/mL (48.94 mM)
形态Liquid
颜色Colorless to light yellow
气味 (Odor)at 100.00 %. woody dusty amber incense
香型woody
旋光度 (Optical Rotation)[α]20/D 138±2°, c = 1% in ethanol
水溶解性59.998μg/L at 25℃
BRN2207559
化妆品成分功效PERFUMING
InChI1S/C15H24/c1-13(2)8-5-6-12-14(3,4)11-7-9-15(12,13)10-11/h6,11H,5,7-10H2,1-4H3/t11-,15-/m0/s1
InChIKeyCQUAYTJDLQBXCQ-NHYWBVRUSA-N
SMILESCC1(C)CCC=C2C(C)(C)[C@H]3CC[C@@]12C3
LogP5.77 at 25℃
安全数据
警示词警告
危险类别码R10
危险品运输编号UN 2319 3/PG 3
WGK Germany3
TSCATSCA listed
REACH 注册登记Active
危险等级3.2
包装类别III
存储类别3 - Flammable liquids
危险性类别Flam. Liq. 3
异长叶烯价格(试剂级)
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2025/12/22 | HY-N7363 | 异长叶烯 Isolongifolene | 1135-66-6 | 1 mg | 514元 |
| 2025/12/22 | HY-N7363 | 异长叶烯 Isolongifolene | 1135-66-6 | 5mg | 1350元 |
| 2025/12/22 | HY-N7363 | 异长叶烯 Isolongifolene | 1135-66-6 | 10 mM * 1 mLin DMSO | 1485元 |
常见问题列表
生物活性
Isolongifolene ((-)-Isolongifolene) 是一种从 Murraya koenigii 中分离的三环倍半萜烯。Isolongifolene 通过调节 P13K/AKT/GSK-3β 信号通路来减轻鱼藤酮诱导的氧化应激,线粒体功能障碍和细胞凋亡。Isolongifolene 具有抗氧化,抗炎,抗癌和神经保护的特性。体外研究
Isolongifolene (0-50 μM; 26 hours; SH-SY5Y neuroblastoma cells) treatment significantly alleviates Rotenone-induced cytotoxicity in SH-SY5Y cells in a dose-dependent manner.
Isolongifolene (10 μM; 26 hours; SH-SY5Y neuroblastoma cells) treatment attenuates Rotenone-induced apoptosis in SH-SY5Y cells.
Isolongifolene (10 μM; 26 hours; SH-SY5Y neuroblastoma cells) treatment attenuates Rotenone induced toxicity by down-regulating Bax, caspases-3, 6, 8 and 9 expression and up-regulating of Bcl-2 expression. Furthermore regulation of p-P13K, p-AKT and p-GSK-3β expression by Isolongifolene.
Cell Viability Assay
| Cell Line: | SH-SY5Y neuroblastoma cells |
| Concentration: | 0 μM, 1 μM, 2.5 μM, 5 μM, 10 μM, 20 μM and 50 µM |
| Incubation Time: | 26 hours |
| Result: | Significantly alleviated Rotenone-induced cytotoxicity in SH-SY5Y cells. |
Apoptosis Analysis
| Cell Line: | SH-SY5Y neuroblastoma cells |
| Concentration: | 10 µM |
| Incubation Time: | 26 hours |
| Result: | Attenuated Rotenone-induced apoptosis in SH-SY5Y cells. |
Western Blot Analysis
| Cell Line: | SH-SY5Y neuroblastoma cells |
| Concentration: | 10 µM |
| Incubation Time: | 26 hours |
| Result: | Attenuated rotenone induced toxicity by down-regulating Bax, caspases-3, 6, 8 and 9 expression and up-regulating of Bcl-2 expression. Prevented the rotenone-induced decreased phosphorylation of GSK-3β. |
