1231930-25-8
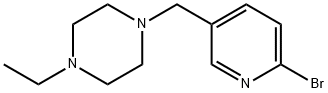 1231930-25-8 结构式
1231930-25-8 结构式
基本信息
1-((6-溴吡啶-3-基)甲基)-4-乙基哌嗪
1-[(6-溴-3-吡啶基)甲基]-4-乙基-哌嗪
1-[(6-broMo-3-pyridinyl)Methyl]-4-ethyl-
1-[(6-Bromo-3-pyridyl)methyl]-4-ethylpiperazine
1-((6-bromopyridin-3-yl)methyl)-4-ethylpiperazine
1-[(6-bromo-3-pyridinyl)methyl]-4-ethylPiperazine
Piperazine, 1-[(6-broMo-3-pyridinyl)Methyl]-4-ethyl-
物理化学性质
制备方法
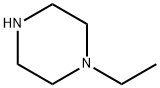
5308-25-8
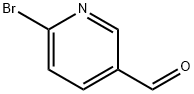
149806-06-4
![1-[(6-溴-3-吡啶基)甲基]-4-乙基-哌嗪](/CAS/20150408/GIF/1231930-25-8.gif)
1231930-25-8
向100 mL无水二氯甲烷(DCM)中加入N-乙基哌嗪(10 g,54 mmol)和2-溴-5-醛基吡啶(6.20 g,54.00 mmol),然后分批加入三乙酰氧基硼氢化钠(NaHB(OAc)3,17.20 g,81.00 mmol)。将反应混合物在室温下搅拌5小时。反应完成后,用DCM(100 mL)稀释混合物,然后加入饱和碳酸钠(Na2CO3)溶液调节pH至8-10。将混合物在水和二氯甲烷之间分配。水相用二氯甲烷萃取两次。合并有机相,依次用水和盐水洗涤,用无水硫酸钠(Na2SO4)干燥,真空浓缩。粗产物通过硅胶柱色谱(洗脱剂:DCM/MeOH = 50:1)纯化,得到目标化合物1-((6-溴吡啶-3-基)甲基)-4-乙基哌嗪(8a,14.00 g,收率92.00%),为黄色固体。质谱(ESI):计算值C12H18BrN3 [M+H]+ 284,实测值284。
参考文献:
[1] Bioorganic and Medicinal Chemistry Letters, 2018, vol. 28, # 5, p. 974 - 978
[2] Patent: CN105622638, 2016, A. Location in patent: Paragraph 0220
[3] Patent: CN106608879, 2017, A. Location in patent: Paragraph 0142-0145
[4] European Journal of Medicinal Chemistry, 2018, vol. 144, p. 1 - 28
[5] Journal of Medicinal Chemistry, 2017, vol. 60, # 5, p. 1892 - 1915
