127369-30-6
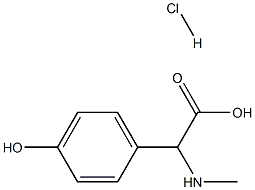 127369-30-6 结构式
127369-30-6 结构式
基本信息
L-对羟基苯甘氨酸甲酯
L-对羟基苯基甘氨酸甲酯
L-对羟基苯甘氨酸甲酯 盐酸盐
(2S)-氨基(4-羟基苯基)乙酸甲酯盐酸盐
(S)-2-氨基-2-(4-羟基苯基)乙酸甲酯盐酸盐
METHYL (2S)-2-AMINO-2-(4-HYDROXYPHENYL)ACETATE HYDROCHLORIDE
L-P-HYDROXYPHENYLGLYCINE METHYL EST
Methyl L-(-)-4-Hydroxyphenylglycinate
Methyl L-(-)-4-Hydroxyphenylglycinate(HCl Salt)
(S)-Methyl 2-amino-2-(4-hydroxyphenyl)acetate HCl
S-4-Hydroxyphenylglycine methyl ester hydrochloride
Methyl (2S)-amino(4-hydroxyphenyl)acetate hydrochloride
Methyl (2S)-amino(4-hydroxyphenyl)ethanoate hydrochloride
Methyl (2S)-2-amino-2-(4-hydroxyphenyl)acetate Hydrochloride
(S)-(2-Amino-2-(4-hydroxy-phenyl)-acetic acid methyl ester hydrochloride
制备方法
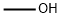
67-56-1
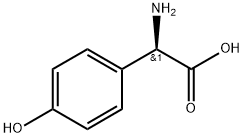
22818-40-2
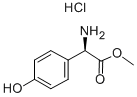
57591-61-4
以甲醇和(R)-2-氨基-2-(4-羟基苯基)乙酸为原料合成D-对羟基苯甘氨酸甲酯盐酸盐的一般步骤:在氮气保护和-15℃条件下,将亚硫酰氯(1.5 mL,33.0 mmol)缓慢滴加到(R)-4-羟基苯基甘氨酸(5.00 g,29.91 mmol)的无水甲醇(25 mL)悬浮液中,同时搅拌。反应混合物逐渐升温至室温并持续搅拌过夜。随后,将黄色溶液回流30分钟。反应混合物经减压浓缩,得到灰白色固体状的D-对羟基苯甘氨酸甲酯盐酸盐。将该盐酸盐在乙醚(15 mL)中搅拌,过滤后用乙醚(2×15 mL)洗涤并干燥,最终得到白色固体状的D-对羟基苯甘氨酸甲酯盐酸盐(6.50 g,100%),其熔点为189-191℃;比旋光度[α]20D = -120.9(c 1, 1M HCl),文献值[α]25D = -121.1(c 1, 1M HCl);红外光谱(KBr)νmax/cm-1:3339(宽峰),1740(C=O),1595;核磁共振氢谱(300 MHz, DMSO-d6)δH:3.68(3H,单峰,OCH3),5.11(1H,单峰,C(2)H),6.80(2H,双峰,J=8.5 Hz,芳香族H),7.25(2H,双峰,J=8.5 Hz,芳香族H),8.70(3H,宽峰,NH3),9.86(1H,单峰,OH)。
参考文献:
[1] Tetrahedron Asymmetry, 2013, vol. 24, # 20, p. 1265 - 1275
[2] European Journal of Medicinal Chemistry, 2018, vol. 145, p. 649 - 660
[3] Synthesis, 2012, vol. 44, # 6, p. 875 - 880
[4] Croatica Chemica Acta, 2003, vol. 76, # 1, p. 23 - 36
[5] Heterocycles, 2009, vol. 78, # 6, p. 1477 - 1483