13343-62-9
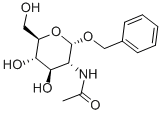 13343-62-9 结构式
13343-62-9 结构式
基本信息
苄基2-乙酰氨基-2-去氧- -D-吡喃葡萄糖苷
苄基-2-乙酰胺基-2-脱氧-Α-D-吡喃葡萄糖苷
苄基 2-乙酰氨基-2-脱氧-Α-D-吡喃葡萄糖苷
苄基-2-O-乙酰氨基-2-脱氧-A-D-葡萄糖苷
N-((2S,3R,4R,5S,6R)-2-(苄氧基)-4,5-二羟基-6-(羟甲基)四氢-2H-吡喃-3-基)乙酰胺
物理化学性质
制备方法
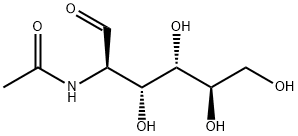
7512-17-6
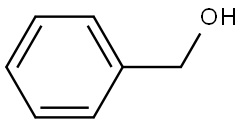
100-51-6

13343-62-9
一般步骤:将N-乙酰氨基葡萄糖(6.40 g,28.9 mmol)溶于苄醇(50 mL,480 mmol)中,加入浓盐酸水溶液(3.0 mL)。将反应混合物在90°C下搅拌3小时。反应完成后,将粗产物冷却至室温,倒入乙醚(500 mL)中,并在4°C下结晶18小时。过滤收集结晶产物,并用石油醚洗涤。通过硅胶柱色谱法(洗脱剂:二氯甲烷/甲醇,95:5至88:12,v/v)纯化,得到苄基糖苷2(6.67 g,21.4 mmol,产率74%),为白色泡沫状固体。Rf = 0.25(二氯甲烷/甲醇,90:10,v/v);熔点182-184°C;[α]D17.8 = +222(c 0.1,甲醇);IR(薄膜)νmax 3298, 3092, 2938, 2901, 2844, 1648, 1552, 1497, 1455, 1375, 1309, 1230, 1156, 1093, 1047, 778, 732, 695 cm?1;1H NMR(500 MHz,CD3OD)δ 7.40-7.25(m,5H,芳烃H),4.85(d,1H,J = 3.6 Hz,H-1′),4.74(d,1H,J = 12.0 Hz,H-1a),4.49(d,1H,J = 12.0 Hz,H-1b),3.89(dd,1H,J = 3.6, 10.8 Hz,H-2′),3.83(dd,1H,J = 1.6, 11.4 Hz,H-6a′),3.73-3.64(m,3H,H-6b′, H-3′, H-4′),3.36(dd,1H,J = 9.6 Hz,H-5′),1.95(s,3H,乙酰基CH3);13C NMR(125 MHz,CD3OD)δ 173.6(C=O,乙酰基),139.0(芳烃季碳),129.4, 129.3, 128.8(芳烃CH),97.5(C-1′),74.1(C-4′),72.7(C-3′),72.5(C-5′),70.1(C-1),62.7(C-6′),55.4(C-2′),22.5(乙酰基CH3);HRMS(ESI)m/z [C15H22NO6]+ 计算值:312.1442,实测值:312.1446。
参考文献:
[1] Journal of Organic Chemistry, 2010, vol. 75, # 10, p. 3515 - 3517
[2] ACS Medicinal Chemistry Letters, 2012, vol. 3, # 3, p. 238 - 242
[3] Chemistry - A European Journal, 2016, vol. 22, # 49, p. 17813 - 17819
[4] Journal of the American Chemical Society, 2012, vol. 134, # 22, p. 9343 - 9351
[5] Bioorganic and Medicinal Chemistry Letters, 2003, vol. 13, # 11, p. 1853 - 1856
