154825-97-5
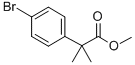 154825-97-5 结构式
154825-97-5 结构式
基本信息
2-(4-溴苯基)-2,2-二甲基乙酸甲酯
甲基2 - (4 - 溴苯基)-2,2 - 二乙酸甲酯
2-(4-BROMOPHENYL)-2,2-DIMETHYLACETATE
P-BROMO-ALPHA,ALPHA-DIMETHYL PHENYLACETIC
methyl 2-(4-bromophenyl)-2-methylpropanoate
P-Bromo-a,a-DimethylPhenylaceticAcidMethylEster
2-(4-bromophenyl)-2-methyl-propionicacidmethyle
α,α-diMethyl-(4-broMophenyl)acetic acid Methyl ester
2-methyl-2-(p-bromophenyl)propionic acid methyl ester
Benzeneacetic acid,4-broMo-a,a-diMethyl-, Methyl ester
Benzeneacetic acid, 4-bromo-α,α-dimethyl-, methyl ester
物理化学性质
制备方法
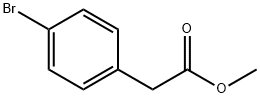
41841-16-1

74-88-4

154825-97-5
在0℃条件下,向干燥的DMF(11 mL)中加入氢化钠(60%油分散,192 mg,4.80 mmol),形成悬浮液。随后,向该悬浮液中缓慢加入2-(4-溴苯基)乙酸甲酯(0.35 mL,2.2 mmol)。将反应混合物在0℃下持续搅拌15分钟后,逐滴加入碘甲烷(6.5 μL,0.10 mmol)。加毕,继续在0℃下搅拌5分钟,然后升至室温搅拌12小时。反应完成后,用1N HCl水溶液(1 mL)淬灭反应,并用乙酸乙酯(3×2 mL)萃取。合并有机层,依次用水(3×2 mL)和饱和食盐水(2 mL)洗涤,无水硫酸钠干燥,过滤后减压浓缩。最后,通过硅胶柱色谱法(洗脱剂:0%至5%乙酸乙酯/己烷梯度洗脱)纯化残余物,得到目标产物2-(4-溴苯基)-2-甲基丙酸甲酯(550 mg,收率98%);质谱(电喷雾正离子模式)C10H11BrO2计算值:242/244,实测值:243/245 [M+H]+。
参考文献:
[1] Patent: WO2014/31928, 2014, A2. Location in patent: Paragraph 0398
[2] Patent: WO2010/86820, 2010, A1. Location in patent: Page/Page column 41-42
[3] Patent: US2010/197591, 2010, A1. Location in patent: Page/Page column 24
[4] Patent: US2008/21024, 2008, A1. Location in patent: Page/Page column 80
[5] Journal of Heterocyclic Chemistry, 2011, vol. 48, # 5, p. 998 - 1005
