168828-82-8
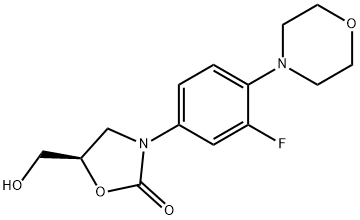 168828-82-8 结构式
168828-82-8 结构式
基本信息
(5R)-3-[3-氟-4-(4-吗啡啉基)苯基]-5-羟甲基-2-恶唑烷酮
(R)-3-(3-FLUORO-4-MORPHOLINOPHENYL)-5-(HYDROXYMETHYL)OXAZOLIDIN-2-ONE
R-N-(3-FLUORO-4-MORPHOLINYLPHENYL)-2-OXO-5-OXAZOLIDINYLMETHANOL
2-Oxazolidinone, 3-[3-fluoro-4-(4-morpholinyl)phenyl]-5-(hydroxymethyl)-, (5R)-
3-(3-FLUORO-4-(4-MORPHOLINYL)PHENYL)-5-HYDROXYMETHYL-(5R)-2-OXAZOLIDIONE
2-Oxazolidinone,3-[3-fluoro-4-(4-morpholinyl)phen
R)-5-(Hydroxymethyl)-3-(3-fluoro-4-morpholinopheny
(5R)-3-[3-Fluoro-4-(4-morpholinyl)phenyl]-5-(hydroxymethyl)-2-oxazolindinone
(5R)-3-(3-FLUORO-4-(4-MORPHOLINYL)PHENYL)-5-HYDROXYMETHYL-2-OXAZOLIDIONE
R-N-[[3-[3-FLUORO-4-MORPHOLINYL]PHENYL]-2-OXO-5-OXAZOLIDINYL]METHANOL
物理化学性质
制备方法
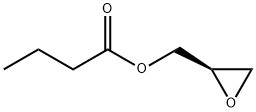
60456-26-0
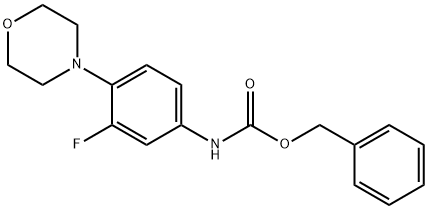
168828-81-7
![(5R)-3-[3-氟-4-(4-吗啡啉基)苯基]-5-羟甲基-2-恶唑烷酮](/CAS/GIF/168828-82-8.gif)
168828-82-8
一般步骤:实施例3:(R)-3-[3-氟-4-(4-吗啉基苯基)]-5-羟甲基-2-恶唑烷酮的制备[131]在氮气保护下,向正丁醇(51.5g)和THF(100ml)的混合溶液中,于20-30℃下缓慢加入正丁基锂(1.6M于己烷中,391.7g),保持反应温度在10-20℃。此过程持续45-60分钟。在另一反应器中,于氮气保护下,将N-羧基苯甲氧基-3-氟-4-吗啉基苯胺(100g)溶于THF(500ml)中,于20-30℃下搅拌。随后将混合物冷却至-15至-5℃,缓慢加入前述制备的正丁基锂溶液,保持温度在-15至-5℃反应45-60分钟。然后,向此溶液中缓慢加入(R)-(-)-缩水甘油丁酸酯(48.0g),于-10至-5℃保持1小时。加料完毕后,将反应温度升至8-13℃维持1小时,再升至13-15℃反应4-5小时。反应完成后,加入水(800ml)和乙酸乙酯(300ml)进行分层。有机层经乙酸乙酯-正己烷混合物洗涤后,固体在空气盘式干燥器中于55-60℃下干燥。产量:0.765g,收率85%。[132]
参考文献:
[1] Synthetic Communications, 2010, vol. 40, # 6, p. 789 - 798
[2] Patent: WO2011/77310, 2011, A1. Location in patent: Page/Page column 12
[3] Organic Syntheses, 2005, vol. 81, p. 112 - 120
[4] Patent: WO2013/111048, 2013, A1. Location in patent: Page/Page column 19; 20
[5] Patent: US2015/25236, 2015, A1. Location in patent: Paragraph 0131; 0132
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2025/05/22 | XW1688288282 | (5R)-3-[3-氟-4-(4-吗啡啉基)苯基]-5-羟甲基-2-恶唑烷酮 (5r)-3-(3-fluoro-4-(4-morpholinyl)phenyl)-5-hydroxymethyl-2-oxazolidione | 168828-82-8 | 25G | 402元 |
| 2025/05/22 | XW1688288281 | (5R)-3-[3-氟-4-(4-吗啡啉基)苯基]-5-羟甲基-2-恶唑烷酮 (5r)-3-(3-fluoro-4-(4-morpholinyl)phenyl)-5-hydroxymethyl-2-oxazolidione | 168828-82-8 | 5G | 115元 |
