171408-76-7
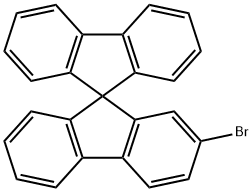 171408-76-7 结构式
171408-76-7 结构式
基本信息
2-溴螺环芴
2-溴-9,9'-螺环芴
2-溴-9,9'-螺二芴
2-溴-9,9'-螺二芴 50G
2-溴-9,9-螺双[9H-芴]
2-溴-9,9'-螺二[9H-芴]
2-溴-9,9'-螺二[9H-芴],98%
2-溴-9,9'-螺二芴 2-BROMO-9,9'-SPIROBIFLUORENE
2- broMospirofluorene
2-BroMo-9,9'-spirobiflu
2-BroMo-9,9'-spirobifluoren
2-BROMO-9,9'-SPIROBIFLUORENE
2-DibroMo-9,9'-spirobifluorene
2-BROMO-9,9''-SPIROBIFLUOROENE
2-Bromo-9,9'-spirobi[9H-fluorene]
9,9′-Spirobi[9H-fluorene],2′-bromo
2-broMo-9,9'-Spirobi[9H]-fluorene (BSBF)
物理化学性质
制备方法
![9-([1,1'-biphenyl]-2-yl)-2-bromo-9H-fluoren-9-ol](/CAS/20180703/GIF/236389-20-1.gif)
236389-20-1

171408-76-7
2.4.1 2-溴-9,9'-螺二芴的合成 在配备温度探针的250 mL三颈烧瓶中,于氮气保护下加入THF(80 mL)和2-溴联苯(9.2 g,40.0 mmol),并安装顶置式搅拌器。将反应体系冷却至-78℃,随后通过注射泵在1小时内缓慢滴加n-BuLi(2.4 M的己烷溶液,20.0 mL)。滴加完毕后,继续在-78℃下搅拌1.5小时。然后,加入2-溴-9-芴酮(10.8 g,45.0 mmol)的THF(100 mL)溶液。移除冰浴,使反应混合物缓慢升温至室温,并通过加入饱和NaHCO3水溶液(200 mL)淬灭反应。用二氯甲烷萃取反应混合物,有机相用无水Na2SO4干燥,随后减压浓缩溶剂,得到黄色粉末状粗产物。 将上述粗产物转移至100 mL三颈烧瓶中,溶于乙酸(40 mL),加入催化量的浓盐酸水溶液(5 mol%,12 N)。将反应混合物回流12小时。冷却至室温后,通过硅胶柱色谱法(洗脱剂:二氯甲烷/正己烷)纯化,得到白色粉末状目标产物2-溴-9,9'-螺二芴(11.3 g,28.8 mmol,收率70%)[36,37]。 1H NMR (CDCl3, 300 MHz): δ 7.83-7.77 (m, 3H), 7.67 (d, J = 8.1 Hz, 1H), 7.45 (dd, J = 8.1, 1.2 Hz, 1H), 7.37-7.30 (m, 3H), 7.11-7.06 (m, 3H), 6.84 (s, 1H), 6.72-6.69 (d, J = 7.5 Hz, 3H). MS (ESI): m/z 434.0 [M + K]+.
参考文献:
[1] Patent: EP2343277, 2011, A2. Location in patent: Page/Page column 44-45
[2] Patent: KR101555155, 2015, B1. Location in patent: Paragraph 0073; 0077
[3] Patent: KR2015/102734, 2015, A. Location in patent: Paragraph 0159-0164
[4] Patent: WO2004/96783, 2004, A1. Location in patent: Page 88
[5] Patent: WO2005/121064, 2005, A1. Location in patent: Page/Page column 32-34
| 报价日期 | 产品编号 | 产品名称 | CAS号 | 包装 | 价格 |
| 2026/06/06 | B3560 | 2-溴-9,9'-螺二[9H-芴] 2-Bromo-9,9'-spirobi[9H-fluorene] | 171408-76-7 | 1g | 110元 |
| 2026/06/06 | B3560 | 2-溴-9,9'-螺二[9H-芴] 2-Bromo-9,9'-spirobi[9H-fluorene] | 171408-76-7 | 5G | 250元 |
| 2025/05/22 | XW88433644101 | 2-(9,9’-螺二[芴]-7-基)-4,4,5,5-四甲基-1,3,2-二氧杂环戊硼烷 2-(9,9’-Spirobi[fluoren]-2-yl)-4,4,5,5-tetramethyl-1,3,2-dioxaborolane | 171408-76-7 | 1g | 41元 |
