478375-42-7
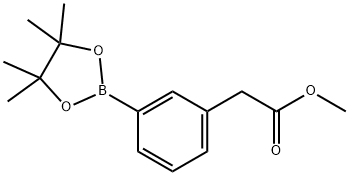 478375-42-7 结构式
478375-42-7 结构式
基本信息
3-(2-甲氧基-2-氧代乙基)苯硼酸频哪醇酯
3-(2-甲氧基-2-氧乙基)苯基硼酸,频哪醇酯
甲基 2(3(4,4,5,5-四甲基-1,3,2-二氧杂硼杂环戊烷-2YL)
Methyl 2-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)
3-(2-Methoxy-2-oxoethyl)phenylboronic acid, pinacol ester
methyl 2-[3-(tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl]acetate
Methyl 2-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenyl)acetate
3-(4,4,5,5-TetraMethyl-1,3,2-dioxaborolan-2-yl)phenyl-acetic acid Methyl ester
Benzeneacetic acid, 3-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-, methyl ester
物理化学性质
制备方法
150529-73-0
73183-34-3

478375-42-7
步骤1:2-(3-(4,4,5,5-四甲基-1,3,2-二氧杂硼杂环戊烷-2-基)苯基)乙酸甲酯的合成 将2-(3-溴苯基)乙酸甲酯(5.00g,21.8mmol)、双(频哪醇合)二硼(11.1g,43.7mmol)和乙酸钾(6.43g,65.5mmol)溶于二恶烷(43.7mL)中,通过鼓泡氮气脱气。加入PdCl2(dppf)-CH2Cl2加合物(0.446g,0.546mmol),将反应混合物加热至85℃并搅拌4小时。反应完成后,用乙酸乙酯(30mL)稀释反应混合物,并通过硅藻土过滤。分离有机层,用盐水洗涤,并用无水硫酸钠干燥。减压蒸发溶剂,残余物通过硅胶柱色谱法纯化(220g硅胶柱,洗脱梯度为0至40%乙酸乙酯/己烷),得到目标产物3-(2-甲氧基-2-氧代乙基)苯硼酸频那醇酯(6.00g,100%),为无色油状物。产物结构经1H NMR(400MHz,CDCl3)确认:δ7.74(s,1H),7.72(s,1H),7.40(t,J=1.7Hz,1H),7.38-7.33(m,1H),3.70(s,3H),3.65(s,2H),1.36(s,12H)。LCMS(M+H)=277.3;HPLC保留时间=0.99分钟(色谱柱:BEH C18 2.1×50mm;流动相A:含0.05%TFA的水;流动相B:含0.05%TFA的乙腈;梯度:2-8%B在1.6分钟内;流速:0.8mL/分钟)。
参考文献:
[1] Patent: WO2015/100282, 2015, A1. Location in patent: Page/Page column 279
[2] Patent: US2016/176864, 2016, A1. Location in patent: Paragraph 0792; 0793
[3] Journal of Medicinal Chemistry, 2007, vol. 50, # 6, p. 1101 - 1115
[4] Patent: US2011/257172, 2011, A1. Location in patent: Page/Page column 19
[5] Patent: US2011/257173, 2011, A1. Location in patent: Page/Page column 19-21
