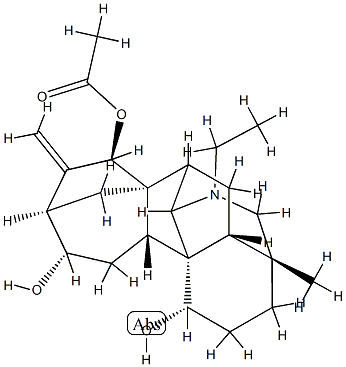
21-Ethyl-4-methyl-16-methylene-7,20-cycloveatchane-1α,12α,15β-triol 15-acetate
- CAS No.
- 5008-49-1
- Chemical Name:
- 21-Ethyl-4-methyl-16-methylene-7,20-cycloveatchane-1α,12α,15β-triol 15-acetate
- Synonyms
- Lucidusculine;21-Ethyl-4-methyl-16-methylene-7,20-cycloveatchane-1α,12α,15β-triol 15-acetate;12,3,6a-Ethanylylidene-9,11a-methanoazuleno[2,1-b]azocine-6,8,11-triol, 1-ethyltetradecahydro-3-methyl-10-methylene-, 11-acetate, (3R,6S,6aR,6bR,8S,9R,11R,11aR,12R,12aR,14R)-
- CBNumber:
- CB72346989
- Molecular Formula:
- C24H35NO4
- Molecular Weight:
- 401.54
- MDL Number:
- MOL File:
- 5008-49-1.mol
21-Ethyl-4-methyl-16-methylene-7,20-cycloveatchane-1α,12α,15β-triol 15-acetate Chemical Properties,Uses,Production
Description
This atisine alkaloid, obtained from Aconitum lucidusculum and A. yeosoensis, was originally given the empirical formula C24H3704N, later changed to that given above. The base crystallizes in colourless leaflets from MeOH and has [α]D- 95.5° (CHCI3). All of the salts crystallize well: the hydrochloride monohydrate, m.p. 98-115°C or 245-265°C (dry, dec.); the hydrobromide, m.p. 248-2500C; [α]20D - 62.7° (H20); the hydriodide as orthorhombic crystals, m.p. 249-2500C; perchlorate decomposing at 260-5°C; [α]15D - 70.3° (EtOH); methiodide, m.p. 197°C; [α]15D - 65° (EtOH) and the picrate, m.p. 173-6°C. The alkaloid contains a secondary and a tertiary hydroxyl group and forms the monoacetate as the crystalline trihydrate, m.p. 113-7°C or 139-144°C (dry); [α]15D- 50.4° (H20). On alkaline hydrolysis, the alkaloid yields acetic acid and Luciduline (q.v.).
References
Majima, Morio.,Proc. Imp. Acad. Tokyo, 7,351 (1931)
Majima, Morio., Ber., 65, 599 (1932)
Suginome, Amiya, Shima., Bull. Chem. Soc., Japan, 32, 824 (1959) V
Crystal structure:
Yoshino, Iitaka., Acta Cryst., 21,57 (1966)