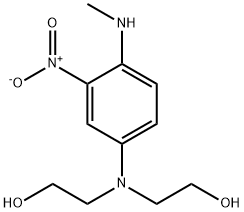
2,2'-((4-(메틸아미노)-3-나이트로페닐)이미노)디에탄올 C화학적 특성, 용도, 생산
일반 설명
Dark blue microcrystals or dark purple powder.
공기와 물의 반응
2,2'-[[4-(methylamino)-3-nitrophenyl]imino]bisethanol may be sensitive to prolonged exposure to air. Slow oxidation. Insoluble in water.
반응 프로필
A nitrated aromatic, amine and alcohol. Amines are chemical bases. They neutralize acids to form salts plus water. These acid-base reactions are exothermic. The amount of heat that is evolved per mole of amine in a neutralization is largely independent of the strength of the amine as a base. Amines may be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen is generated by amines in combination with strong reducing agents, such as hydrides. Flammable and/or toxic gases are generated by the combination of alcohols with alkali metals, nitrides, and strong reducing agents. They react with oxoacids and carboxylic acids to form esters plus water. Oxidizing agents convert them to aldehydes or ketones. Alcohols exhibit both weak acid and weak base behavior. They may initiate the polymerization of isocyanates and epoxides.
화재위험
Flash point data for 2,2'-[[4-(methylamino)-3-nitrophenyl]imino]bisethanol are not available; however 2,2'-[[4-(methylamino)-3-nitrophenyl]imino]bisethanol is probably combustible.
2,2'-((4-(메틸아미노)-3-나이트로페닐)이미노)디에탄올 준비 용품 및 원자재
원자재
준비 용품