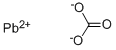LEAD CARBONATE suppliers
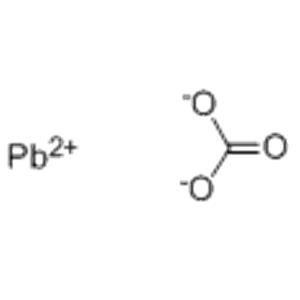
LEAD CARBONATE
- CAS:
- 598-63-0
- MF:
- CO3Pb
- MW:
- 267.21
Suppliers by country/region
Company Type
Properties
- Melting point:
- 399-401°C (dec.)
- bulk density
- 1300kg/m3
- Density
- 6.6 g/cm3
- refractive index
- 2.08
- storage temp.
- Store below +30°C.
- solubility
- insoluble in H2O
- form
- Powder
- color
- White
- PH
- 5.8 (50g/l, H2O, 20℃)(slurry)
- Water Solubility
- Soluble in acid and alkali. Insoluble in water, alcohol and ammonia.
- Dielectric constant
- 18.1(16.0℃)
- Stability:
- Stable. Incompatible with strong acids, strong oxidizing agents.
- InChI
- 1S/CH2O3.Pb/c2-1(3)4;/h(H2,2,3,4);/q;+2/p-2
- InChIKey
- MFEVGQHCNVXMER-UHFFFAOYSA-L
- SMILES
- [Pb+2].[O-]C(=O)[O-]
- CAS DataBase Reference
- 598-63-0(CAS DataBase Reference)
- EPA Substance Registry System
- Lead(II) carbonate (598-63-0)
Safety Information
- Symbol(GHS)



GHS07,GHS08,GHS09
- Signal word
- Danger
- Hazard statements
- H302+H332-H360Df-H373-H410
- Precautionary statements
- P202-P260-P273-P301+P312-P304+P340+P312-P308+P313
- Hazard Codes
- T,N
- Risk Statements
- 61-20/22-33-50/53-62-45
- Safety Statements
- 53-45-60-61
- RIDADR
- UN 3077 9/PG 3
- WGK Germany
- 3
- RTECS
- OF9275000
- TSCA
- TSCA listed
- HS Code
- 2836 99 17
- HazardClass
- 6.1
- PackingGroup
- III
- Storage Class
- 6.1D - Non-combustible, acute toxic Cat.3
toxic hazardous materials or hazardous materials causing chronic effects
- Hazard Classifications
- Acute Tox. 4 Inhalation
Acute Tox. 4 Oral
Aquatic Acute 1
Aquatic Chronic 1
Repr. 1A
STOT RE 2
- Hazardous Substances Data
- 598-63-0(Hazardous Substances Data)
- Limited Quantities
- 5.0 L (1.3 gallons) (liquid) or 5.0 kg (11 lbs) (solid)
- Excepted Quantities
- Max Inner Pack (30g or 30ml) and Max Outer Pack (1Kg or 1L)
Use
When heated at 315C, lead carbonate decomposes to lead oxide and carbon dioxide: PbCO3PbO + CO2 When heated in water, it transforms to basic lead carbonate, 2PbCO3Pb(OH)2 3PbCO3 + H2O 2PbCO3Pb(OH)2 + CO2 Lead carbonate dissolves in acids, forming the corresponding lead salt and evolving carbon dioxide: PbCO3 + 2HCl PbCl2 + H2O + CO2 Reaction with concentrated acetic acid yields anhydrous lead(II) acetate. Fusion with boric acid at high temperature forms lead metaborate that has an approximate composition Pb(BO2)2H2O. The product loses water of crystallization at 160C.
143 supplier list of "LEAD CARBONATE"
- Product Name:LEAD CARBONATE
- Products Intro:Purity: 99% | Package: 1KG
- Company Type: Trader
- Country/Region: CHINA
- Product Name:Lead(II)carbonate
- Company Type: Reagent
- Country/Region: CHINA
- Main Products: Gamma-butyrolactone
- Product Name:Lead(II) carbonate
- Products Intro:Purity: 0.99
- Company Type: Trader
- Country/Region: CHINA
- Main Products: Fine Chemical,API,Raw Material,Pharmaceutical Compounds,Others
- Product Name:Lead(II) carbonate
- Products Intro:Purity: 0.95&0.99 | Package: 0.1KG;1KG;1000KG | CustNote: Hot sales
- Company Type: Trader
- Country/Region: CHINA
- Main Products: Triglycol monomethyl ether, CAS 112-35-6,Tetrafluoro-1-propanol, CAS 76-37-9,Aminoacetaldehyde dimethyl acetal, CAS 22483-09-6,Hexachloroethane, CAS 67-72-1,3-Allyloxy-2-Hydroxy-1-Propane,Sodium Salt, CAS 52556-42-0
You can find LEAD CARBONATE suppliers,
manufacturers, and distributors from countries such as China, United States and the United Kingdom here.
Browse to the tetrahydrofuran product information displayed by the supplier,
as well as the supplier's contact and capability information.