|
|
| | Hydroxychloroquine Sulfate EP Impurity A Basic information |
| Product Name: | Hydroxychloroquine Sulfate EP Impurity A | | Synonyms: | Hydroxychloroquine Sulfate EP Impurity A;Hydroxychloroquine EP Impurity ABCDEFG;Hydroxychloroquine Sulfate Impurity 5(Hydroxychloroquine Sulfate EP Impurity A);Ethanol, 2-[[4-[(7-chloro-4-quinolinyl)amino]pentyl]ethyloxidoamino]-;4-[(7-Chloroquinolin-4-yl)amino]-N-ethyl-N-(hydroxyethyl)pentan-1-amineN-oxide (Mixture of diastereoisomers);4-((7-chloroquinolin-4-yl)amino)-N-ethyl-N-(2-hydroxyethyl)pentan-1-amine oxide;Hydroxychloroquine EP Impurity A / Hydroxychloroquine N-Oxide;Hydroxychloroquine sulfate Impurity A | | CAS: | 1449223-88-4 | | MF: | C18H26ClN3O2 | | MW: | 351.88 | | EINECS: | | | Product Categories: | | | Mol File: | 1449223-88-4.mol | 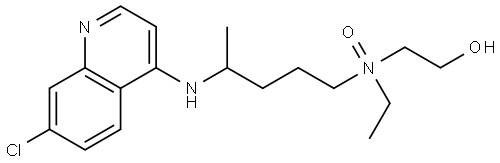 |
| | Hydroxychloroquine Sulfate EP Impurity A Chemical Properties |
| solubility | Chloroform (Slightly), Methanol (Slightly) | | pka | 13.71±0.10(Predicted) | | form | Sticky Solid to Solid | | color | Light Yellow to Yellow | | Stability: | Very Hygroscopic |
| | Hydroxychloroquine Sulfate EP Impurity A Usage And Synthesis |
| Uses | Hydroxychloroquine N-Oxide is a derivative of Chloroquine (C379965), a standard anti-malarial drug. Substrate for MRP in multidrug resistant cell line and inhibits photoaffinity labelling of MRP by quinoline-based photoactive drug IAAQ. It is a COVID19-related research product. |
| | Hydroxychloroquine Sulfate EP Impurity A Preparation Products And Raw materials |
|