|
|
| | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester Basic information |
| Product Name: | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester | | Synonyms: | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester;(E)-3-hydroxy-2,3-dimethylbutan-2-yl hydrogen 2-ethoxyvinylboronate;E-2-Ethoxyethenyl-1-boronic acid pinacol ester;trans-2-Ethoxyethenyl-1-boronic acid pinacol ester≥ 98%;trans-2-Ethoxyvinylboronic acid pinacol ester 97%;(E)-2-Ethoxyvinylboronic acid pinacol ester;trans-2-Ethoxyvinylboronic acid pinacol ester;1,3,2-Dioxaborolane, 2-[(1E)-2-ethoxyethenyl]- 4,4,5,5-tetraMethyl- | | CAS: | 1201905-61-4 | | MF: | C10H19BO3 | | MW: | 198.07 | | EINECS: | | | Product Categories: | | | Mol File: | 1201905-61-4.mol | 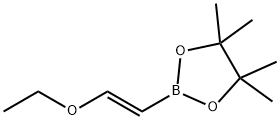 |
| | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester Chemical Properties |
| Boiling point | 50℃/0.3mm | | density | 0.935 g/mL at 25 °C | | refractive index | 1.4460 | | Fp | 86℃ | | storage temp. | 2-8°C | | form | liquid | | color | Clear, colourless | | InChIKey | MRAYNLYCQPAZJN-BQYQJAHWSA-N |
| RIDADR | NA 1993 / PGIII | | WGK Germany | 3 | | HS Code | 2931900090 |
| | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester Usage And Synthesis |
| Uses | trans-2-Ethoxyvinylboronic acid pinacol ester is a boronic ester commonly used in Suzuki-Miyaura cross-coupling.
This reaction is a key step to synthesize:
- Azaindole and diazaindoles from chloroamino-N-heterocycles.
- Doryanine and its derivatives from 2-bromobenzoic acid.
|
| | (E)-1-Ethoxyethene-2-ylboronic acid pinacol ester Preparation Products And Raw materials |
|