|
|
| | Enasidenib Basic information |
| Product Name: | Enasidenib | | Synonyms: | Enasidenib(AG-221);AG-221 (Enasidenib);AG-211;2-Methyl-1-(4-(6-(trifluoromethyl)pyridin-2-yl)-6-(2-(trifluoromethyl)pyridin-4-ylamino)-1,3,5-triazin-2-ylamino)propan-2-ol;Enasidenib;AG-221;2-Propanol, 2-methyl-1-[[4-[6-(trifluoromethyl)-2-pyridinyl]-6-[[2-(trifluoromethyl)-4-pyridinyl]amino]-1,3,5-triazin-2-yl]amino]-;Enasidenib Mesylate | | CAS: | 1446502-11-9 | | MF: | C19H17F6N7O | | MW: | 473.38 | | EINECS: | | | Product Categories: | Inhibitors | | Mol File: | 1446502-11-9.mol | 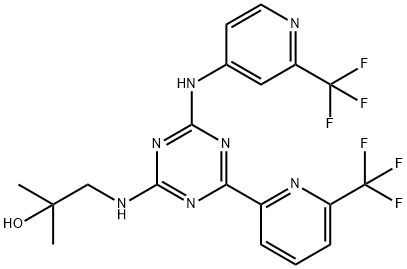 |
| | Enasidenib Chemical Properties |
| Melting point | 168-170°C | | Boiling point | 581.0±60.0 °C(Predicted) | | density | 1.477±0.06 g/cm3(Predicted) | | storage temp. | -20°C | | solubility | Soluble in DMSO (up to 25 mg/ml) | | form | solid | | pka | 14.70±0.29(Predicted) | | color | White | | Stability: | Stable for 1 year from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months. |
| | Enasidenib Usage And Synthesis |
| Description | Enasidenib (1446502-11-9) is a potent (IC50’s = 100 nM IDH2R140Q homodimer, 30 nM IDH2R140Q/WT heterodimer and 10 nM IDH2R172K/WT heterodimer) and selective inhibitor of mutant isocitrate dehydrogenase 2 (IDH2).1? It suppressed the production of the oncometabolite (R)-2-Hydroxyglutarate (a competitive inhibitor of αKG-dependent dioxygenases which leads to epigenetic dysregulation) and induced cellular differentiation in primary human IDH2 mutation-positive acute myeloid leukemia cells.1,2? Recently approved for clinical use by the FDA. | | Uses | Enasidenib is a first-in-class oral selective inhibitor of mutant IDH2 enzymes (isocitrate dehydrogenase 2), for the treatment of adults with relapsed or refractory IDH2-mutated acute myeloid leukemia. | | Definition | ChEBI: Enasidenib is a 1,3,5-triazine which is substituted by (2-hydroxy-2-methylpropyl)nitrilo, 6-(trifluoromethyl)pyridin-2-yl and [2-(trifluoromethyl)pyridin-4-yl]nitrilo groups at positions 2,4 and 6, respectively. It is an isocitrate dehydrogenase-2 (IDH2) inhibitor which has been approved for the treatment of adults with relapsed or refractory acute myeloid leukaemia (AML). It has a role as an antineoplastic agent and an EC 1.1.1.42 (isocitrate dehydrogenase) inhibitor. It is an aminopyridine, an organofluorine compound, a secondary amino compound, a tertiary alcohol, a member of 1,3,5-triazines and an aromatic amine. | | in vitro | ag-221 was found to be able to reduce 2-hg levels by >90%, reverse in-vitro histone and dna hypermethylation, and induce differentiation in leukemia cell model as well. in addition, a dose dependent proliferative burst of the human specific cd45+ blast cells was observed by the treatment of ag-221, as measured by the expression of cd11b, cd14, cd15 and cell morphology [1]. | | in vivo | the efficacy of ag-221 in a primary human aml xenograft model with the idh2 r140q mutation was studied, and the results showed that ag-221 could reduce 2-hg in the plasma, bone marrow, and urine of engrafted mice potently. in addition, the treatment of ag-221 could also induce a significant and dose dependent survival benefit as demonstrated by that all mice in the high dose treatment of ag-221 survived to the end of study [1]. | | IC 50 | ~16 nm for idh2 r140q mutant | | References | 1) Yen et al. (2017), AG-221, A First-in-Class Therapy Targeting Acute Myeloid Leukemia Harboring Oncogenic IDH2 Mutations; Cancer Discov. 7 478
2) Amatangelo et al. (2017), Enasidenib induces acute myeloid leukemia cell differentiation to promote clinical response; Blood 130 732 |
| | Enasidenib Preparation Products And Raw materials |
|