|
|
| | Azolimine Basic information |
| Product Name: | Azolimine | | Synonyms: | 2-Imino-2,3,4,5-tetrahydro-3-methyl-1-phenyl-1H-imidazol-4-one;2-Imino-3-methyl-1-phenyl-4-imidazolidinone;CL-90748;Azolimine;4-Imidazolidinone, 2-imino-3-methyl-1-phenyl- | | CAS: | 40828-45-3 | | MF: | C10H11N3O | | MW: | 189.21 | | EINECS: | | | Product Categories: | | | Mol File: | 40828-45-3.mol | 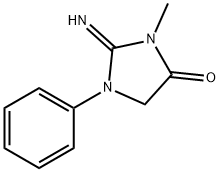 |
| | Azolimine Chemical Properties |
| Boiling point | 265.0±23.0 °C(Predicted) | | density | 1.25±0.1 g/cm3(Predicted) | | pka | 2.81±0.30(Predicted) |
| | Azolimine Usage And Synthesis |
| Originator | Azolimine,ZYF Pharm Chemical | | Uses | Diuretic. | | Manufacturing Process | 2 methods of producing of 1-phenyl-2-imino-3-methyl-4-oxoimidazolidine:
1). In 29 ml of water containing 1.0 g of sodium hydroxide was dissolved 3.55
g of phenylcyanamide. To the clear solution was added 2.7 g of Nmethylchloroacetamide.
The reaction mixture was left to stir for 120 h at
room temperature. The solvent was therefore stripped off on a rotary
evaporator under vacuum. The solid residue was slurried in dilute alkali and
filtered to yield 1.0 g (21%) of 1-phenyl-2-imino-3-methyl-4-oxoimidazolidine,
melting point 163°-165°C.
2). 133.0 g of phenylcyanamide hemihydrate was dissolved in 1500 ml of
absolute alcohol. To this solution was added 55.0 g of sodium methoxide. This
mixture was stirred for 30 min and 113.0 g of N-methylchloroacetamide then
added. This mixture was heated to reflux for 4 h and allowed to stand
overnight. The white precipitate was filtered and washed with ethanol. This
ethanol filtrate was concentrated on the rotating evaporator and cooled giving
additional product. Both crops of product were combined, washed with water
and dried to give 87.0 g (47%) of 1-phenyl-2-imino-3-methyl-4-
oxoimidazolidine, melting point 169°-170°C. | | Therapeutic Function | Diuretic |
| | Azolimine Preparation Products And Raw materials |
|