|
|
| | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt Basic information |
| Product Name: | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt | | Synonyms: | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt;N-(Propargyl-peg4)-n-bis(peg4-acid);QUN 53092;4,7,10,13,19,22,25,28-Octaoxa-16-azahentriacontanedioic acid, 16-(3,6,9,12-tetraoxapentadec-14-yn-1-yl)- | | CAS: | 2093153-09-2 | | MF: | C33H61NO16 | | MW: | 727.83 | | EINECS: | | | Product Categories: | | | Mol File: | 2093153-09-2.mol | 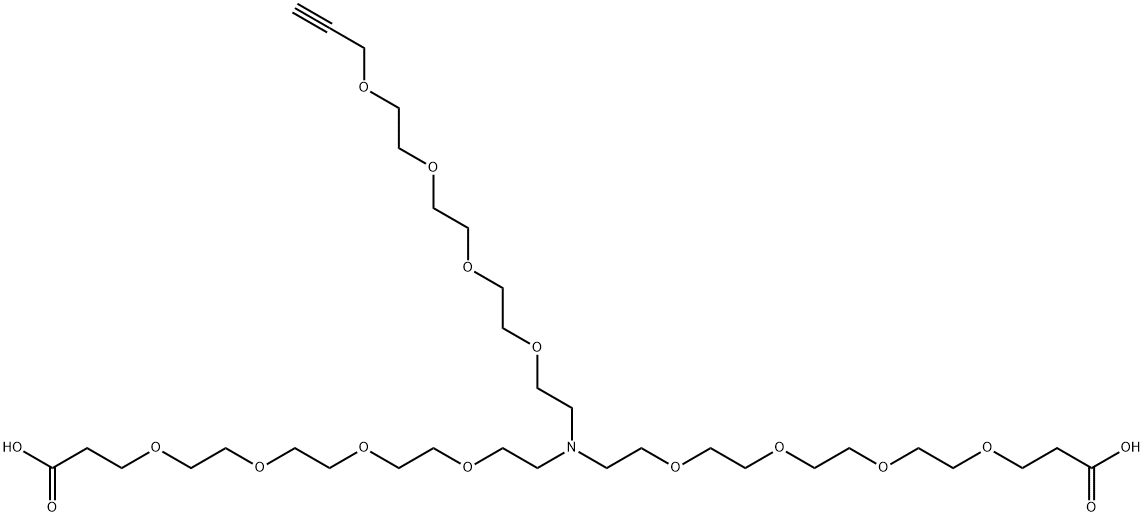 |
| | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt Chemical Properties |
| Boiling point | 765.0±60.0 °C(Predicted) | | density | 1.155±0.06 g/cm3(Predicted) | | solubility | Soluble in Water, DMSO, DCM, DMF | | pka | 3.97±0.10(Predicted) |
| | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt Usage And Synthesis |
| Description | N-(Propargyl-PEG4)-N-bis(PEG4-acid) is a branched crosslinking reagent with a propargyl group and two terminal carboxylic acids. The propargyl group can react with azide-bearing compounds or biomolecules via copper catalyzed azide-alkyne Click Chemistry to yield a stable triazole linkage. The terminal carboxylic acids can react with primary amino groups in the presence of activators (e.g. EDC, or HATU) to form a stable amide bond. |
| | N-(Propargyl-PEG4)-N-bis(PEG4-acid) HCl salt Preparation Products And Raw materials |
|