|
|
| | (S) QuinoxP(R) Basic information | | Reaction |
| Product Name: | (S) QuinoxP(R) | | Synonyms: | (S) QuinoxP;QuinoxP;(S,S)-QuinoxP*;2,3-Bis[(S)-(1,1-dimethylethyl)methylphosphino]quinoxaline;(S,S)-(+)-2,3-Bis(t-butylmethylphosphino)quinoxaline;(S) QuinoxP(R);(S,S)-2,3-Bis(tert-butylmethylphosphino)quinoxaline;(S,S)-(+)-2,3-Bis(t-butylMethylphosphino)quinoxaline (S,S)-QuinoxP* | | CAS: | 1107608-80-9 | | MF: | C18H28N2P2 | | MW: | 334.38 | | EINECS: | | | Product Categories: | Chiral Phosphine | | Mol File: | 1107608-80-9.mol | 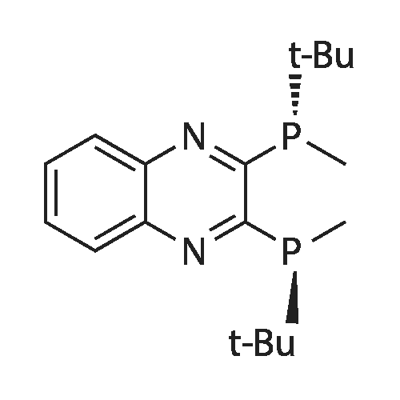 |
| | (S) QuinoxP(R) Chemical Properties |
| Melting point | 100-105 °C | | Boiling point | 447.6±45.0 °C(Predicted) | | alpha | +54.3° (c 1.0, CHCl3) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | pka | -0.54±0.59(Predicted) | | form | Powder | | color | orange | | optical activity | [α]22/D +55°, c = 1 in chloroform |
| Hazard Codes | Xn | | Risk Statements | 22-36/37/38 | | Safety Statements | 26 | | RIDADR | UN 2811 6.1/PG 3 | | WGK Germany | 3 | | HS Code | 2933.99.8290 |
| | (S) QuinoxP(R) Usage And Synthesis |
| Reaction |
- Ligand for the rhodium-catalyzed, asymmetric hydrogenation of dehydroamino acid esters and α-enamides.
- Ligand for the rhodium-catalyzed, asymmetric 1,4-addition of arylboronic acids to α,β-unsaturated carbonyl compounds.
- Ligand for the rhodium-catalyzed, asymmetric alkylative ring opening reaction
- Ligand for the palladium-catalyzed asymmetric allylic alkylation and amination of racemic substrates.
- Ligand for the ruthenium-catalyzed asymmetric hydrogenation of ketones.
- Ligand for the rhodium-catalyzed, asymmetric hydroacylation of 1,1-disubstituted alkenes with aldehydes.
- Ligand for the silver-catalyzed asymmetric nitroso aldol reaction.
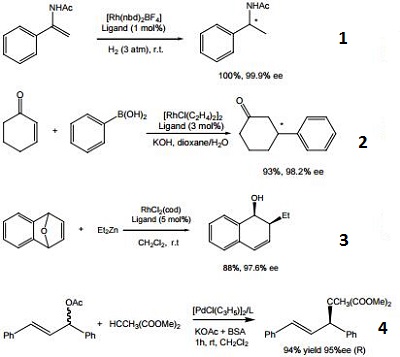
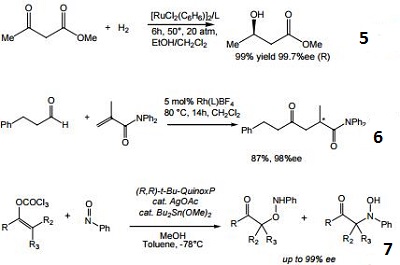
| | Uses | (S,S)-(+)-2,3-Bis(tert-butylmethylphosphino)quinoxaline is a useful reagent for organic synthesis of chiral tetraphenylenes, fused lactones and other useful intermediates. |
| | (S) QuinoxP(R) Preparation Products And Raw materials |
|