
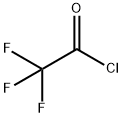
TRIFLUOROACETYL CHLORIDE
| Price | $1 |
| Package | 1KG |
| Min. Order: | 1KG |
| Supply Ability: | 1ton |
| Update Time: | 2019-12-31 |
Product Details
| Product Name: TRIFLUOROACETYL CHLORIDE | CAS No.: 354-32-5 |
| EC-No.: 200-158-5 | Min. Order: 1KG |
| Purity: 95-99% | Supply Ability: 1ton |
| Release date: 2019/12/31 | |
| Contact: Flora@coreychem.com |
▼
▲
TRIFLUOROACETYL CHLORIDE Basic information
▼
▲
Product Name:
TRIFLUOROACETYL CHLORIDE
Synonyms:
trifluoro-acetylchlorid;TRIFLUOROACETYL CHLORIDE CYL. WITH 800;Trifluoroacetylchloride97%;Trifluoracetylchlorid;Trifluoroacetic acid chloride;2,2,2-trifluoroacetyl chloride;2,2,2-trifluoroethanoyl chloride;TRIFLUOROACETYL CHLORIDE
CAS:
354-32-5
MF:
C2ClF3O
MW:
132.47
EINECS:
206-556-2
Product Categories:
Chemical Synthesis;Compressed and Liquefied Gases;Synthetic Reagents;Chemical Synthesis;Specialty Gases;Synthetic Reagents;-
Mol File:
354-32-5.mol
▼
▲
TRIFLUOROACETYL CHLORIDE Chemical Properties
▼
▲
Melting point
−146 °C(lit.)
Boiling point
−27 °C(lit.)
density
1,335 g/cm3
vapor density
4.6 (vs air)
Fp
-58.1±25.9℃
BRN
1098994
CAS DataBase Reference
354-32-5(CAS DataBase Reference)
EPA Substance Registry System
Trifluoroacetyl chloride (354-32-5)
▼
▲
Safety Information
▼
▲
Hazard Codes
C
Risk Statements
14-35-37
Safety Statements
26-36/37/39-45
RIDADR
UN 3057 2.3
WGK Germany
2
RTECS
AO7150000
F
4.5-19-21
Hazard Note
Corrosive
HazardClass
2.3
▼
▲
MSDS Information
▼
▲
Provider
Language
SigmaAldrich
English
▼
▲
TRIFLUOROACETYL CHLORIDE Usage And Synthesis
▼
▲
Uses
Widely used in medicine, pesticides, organic intermediate and fine chemical.
General Description
A colorless gas. Shipped as a liquid under own vapor pressure. Contact with the unconfined liquid may frostbite unprotected skin. Very toxic by inhalation and may severely irritate skin, eyes, and mucous membranes. Under prolonged exposure to fire or heat the containers may rupture violently and rocket.
Air & Water Reactions
Reacts avidly with water and with moisture in the air to give fumes of hydrogen chloride, a water-soluble toxic gas.
Reactivity Profile
TRIFLUOROACETYL CHLORIDE is incompatible with strong oxidizing agents, alcohols, amines, alkalis. Reacts vigorously with amines and alkalis. May react vigorously or explosively if mixed with diisopropyl ether or other ethers in the presence of trace amounts of metal salts [J. Haz. Mat., 1981, 4, 291].
Health Hazard
TOXIC; may be fatal if inhaled, ingested or absorbed through skin. Vapors are extremely irritating and corrosive. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.
Fire Hazard
Some may burn but none ignite readily. Vapors from liquefied gas are initially heavier than air and spread along ground. Some of these materials may react violently with water. Cylinders exposed to fire may vent and release toxic and/or corrosive gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.
Safety Profile
Corrosive to skin, eyes, and materials. When heated to decomposition it emits very toxic fumes of Fand Cl-. See also FLUORIDES and CHLORIDES.
Company Profile Introduction
Established in 2014,Career Henan Chemical Co. is a manufacturerspecializing in the sale of fine chemicals.
Mainly deals in the sales of:
Pharmaceutical intermediates
OLED intermediates:
Pharmaceutical intermediates;
OLED intermediates;
You may like
Recommended supplier
| Product name | Price | Suppliers | Update time | |
|---|---|---|---|---|
| $10.50/1KG |
VIP5Y
|
Hebei Chuanghai Biotechnology Co,.LTD
|
2024-08-21 | |
| $0.00/1Kg |
VIP7Y
|
Shaanxi Dideu Medichem Co. Ltd
|
2020-01-06 | |
| $0.00/5mg |
VIP4Y
|
TargetMol Chemicals Inc.
|
2026-01-05 | |
| $2.00/100kg |
VIP1Y
|
ZHENGZHOU JIUYI TIME NEW MATERIALS CO,.LTD
|
2025-06-25 | |
| $0.00/1G |
VIP1Y
|
Shenzhen Nexcon Pharmatechs Ltd.
|
2025-06-03 |
- Since: 2014-12-17
- Address: 702, Building 10, East District, National University Science and Technology Park, High tech Zone, Zh
INQUIRY



![BENZO[D]ISOXAZOL-3-YL-METHANESULFONYL CHLORIDE](https://img.chemicalbook.com/ProductImageEN/2020-1/Large/034910f9-08bc-48f7-9a8c-c021cbfe2c57.png)

 China
China