Product Details
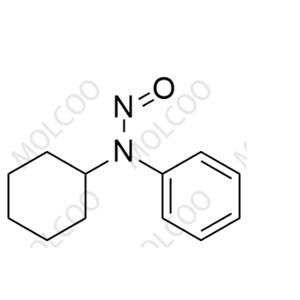
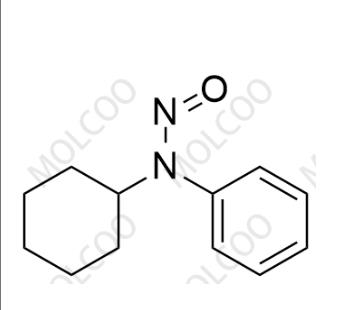
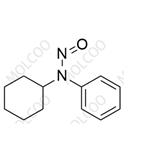
| Product Name: N-cyclohexyl-N-phenylnitrous amide | CAS No.: 54955-24-7 |
| Min. Order: 10mg | Purity: 99%+ HPLC |
| Supply Ability: 1000 | Release date: 2025/07/31 |
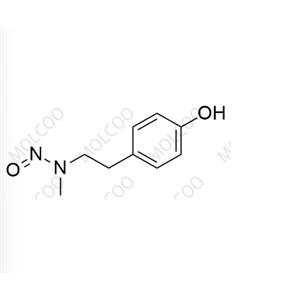
N-cyclohexyl-N-phenylnitrous amide;54955-24-7
Product Information
Advantages
Applications
Background Description
Research Status
Company Profile Introduction
-
You may like
Recommended supplier
| Product name | Price | Suppliers | Update time | |
|---|---|---|---|---|
| $39.00/2mg |
VIP6Y
|
TargetMol Chemicals Inc.
|
2025-11-05 | |
| $1.00/1g |
VIP5Y
|
RongNa Biotechnology Co.,Ltd
|
2025-08-22 | |
| $0.00/200kg |
VIP2Y
|
Qingdao RENAS Polymer Material Co., Ltd.
|
2025-03-24 |
INQUIRY








 China
China