Zonisamide Impurity
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
Product Information
Product Number: Z010008
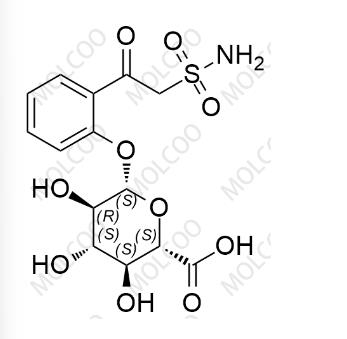
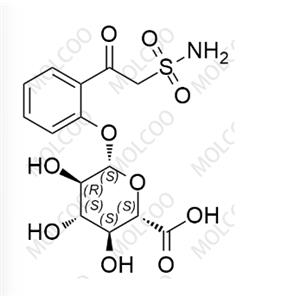
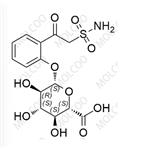
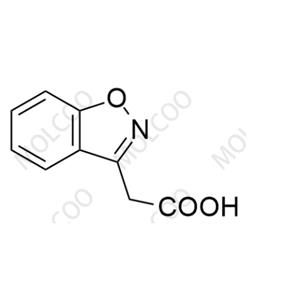
English Name: Zonisamide Impurity 8
English Alias: (2S,3S,4S,5R,6S)-3,4,5-trihydroxy-6-(2-(2-sulfamoylacetyl)phenoxy)tetrahydro-2H-pyran-2-carboxylic acid
CAS Number: 84557-58-4
Molecular Formula: C14H17NO10S
Molecular Weight: 391.35
Advantages: As a reference standard for Zonisamide Impurity 8, it has a clear chemical structure, and its purity is strictly determined (≥98%). It has good stability and uniformity, remaining stable under various storage conditions (such as in a light-protected, low-temperature environment) without easy decomposition or deterioration. It can serve as a reliable reference substance, ensuring the accuracy and repeatability of impurity detection results for zonisamide bulk drugs and formulations, meeting the strict requirements of drug research, development, production, and quality supervision, and providing solid support for drug quality control.
Applications:
Development of Quality Testing Methods: Used to establish and validate detection methods for Impurity 8 in zonisamide, such as high-performance liquid chromatography (HPLC) and liquid chromatography - mass spectrometry (LC - MS), determining detection conditions and parameters to achieve precise quantitative analysis of impurities.
Production Process Monitoring: During the production of zonisamide, monitor the content of Impurity 8 in real time. Optimize the synthesis process (such as reaction temperature, time, raw material ratio, etc.) to reduce impurity generation and ensure that product quality meets standards.
Stability Studies: In drug stability tests (accelerated tests, long-term tests), track the changes in the content of Impurity 8, evaluate the quality stability of drugs under different storage conditions, and provide data support for determining the shelf life and storage conditions of drugs.
Regulatory Compliance: Meet the requirements of domestic and international drug regulatory agencies (such as FDA, EMA, NMPA) for drug impurity limits, and assist enterprises in drug registration and marketing applications.
Background Description: Zonisamide is a drug used in the treatment of neurological diseases such as epilepsy. During its research, development, and production, the presence of impurities may affect the safety, effectiveness, and stability of the drug. To ensure patient safety and comply with drug regulatory requirements, strict research and precise control of impurities in zonisamide are essential. As one of the important impurities of zonisamide, in-depth research on Zonisamide Impurity 8 helps to improve the quality standard system of zonisamide, enhance drug quality, and ensure the safety and effectiveness of clinical medication.
Research Status:
Detection Technology: Currently, the HPLC - UV method is mainly used for the detection of Impurity 8. By optimizing conditions such as the type of chromatographic column and the composition of the mobile phase, effective separation and detection of Impurity 8 can be achieved, with a detection limit of up to 0.05%. Meanwhile, the application of LC - MS technology can further improve the sensitivity and accuracy of detection, helping to identify the structure of impurities.
Formation Mechanism: Research has found that Impurity 8 may originate from side reactions of raw materials and incomplete conversion of intermediates during the synthesis of zonisamide. Through in-depth analysis of the synthesis route, the key factors affecting impurity generation have been clarified, providing a theoretical basis for process optimization.
Safety Evaluation: Currently, there are relatively few toxicological studies on Impurity 8. However, preliminary experiments have shown that high concentrations of Impurity 8 may have a certain impact on cell activity. Therefore, strict limits have been set for it in drug quality standards to ensure drug safety.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!









 China
China