| структура |
наименование химического вещества |
Номер CAS |
молекулярная формула |
 |
Beta-cyclodextrin (medicinal excipients) |
|
|
 |
Calcium diphosphate dihydrate (medicinal excipients) |
|
|
 |
Sucralose (medicinal excipients) |
|
|
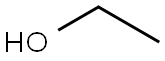 |
Ethanol (95%) (medicinal excipients) |
|
C2H6O |
 |
Total ionic strength regulator PH5.0-5.5 |
|
|
 |
Tartaric acid (medicinal excipients) |
|
|
 |
Methanesulfonic acid (medicinal excipients) |
|
|
 |
DL-tartaric acid (medicinal excipients) |
|
|
 |
Anhydrous sodium acetate (medicinal excipients) |
|
|
 |
Succinic acid (medicinal excipients) |
|
|
 |
Acetic acid-ammonium acetate buffer (pH4.8) |
|
|
 |
Magnesium stearate (medicinal excipients) |
|
|
 |
Dimethicone (medicinal excipients) |
|
|
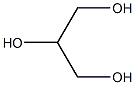 |
Glycerin (for injection) (medicinal excipients) |
|
C3H8O3 |
 |
Hexadecanol (medicinal excipient) |
|
|
 |
Polyvinyl alcohol (medicinal excipients) |
|
|
 |
Beeswax, synthetic |
71243-51-1 |
|
 |
Medicinal yellow beeswax |
|
|
 |
Soy lecithin (medicinal excipients for injection) |
|
|
 |
Tyloxapol (medicinal excipients) |
|
|
 |
Sodium lauryl sulfate (medicinal excipients) |
|
|
 |
Carboxymethyl starch sodium |
|
|
 |
White beeswax (medicinal excipients) |
|
|
 |
Soybean oil (medicinal excipients) |
|
|
 |
TX-10 |
|
|
 |
Capo 940 |
|
|
 |
Sucrose stearate (medicinal excipients) |
|
|
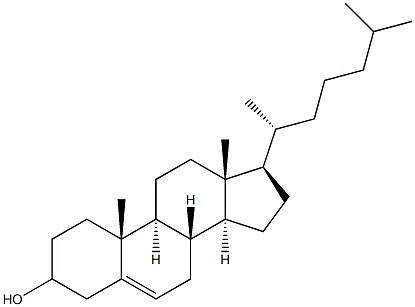 |
Cholesterol (for injection) (medicinal excipients) |
|
C27H46O |
 |
Triethanolamine (medicinal excipients) |
|
|
 |
Methyl cellulose (medicinal excipients) |
|
|
 |
Soybean phospholipid (medicinal excipient) |
|
|
 |
Emulsifier 400 do |
|
|
 |
Hydroxyethyl Cellulose |
|
|
 |
Pharmaceutical grade sodium carboxymethyl starch |
|
|
 |
Egg yolk lecithin (for injection) (medicinal excipients) |
|
|
 |
Polyethylene glycol monomethyl etherified polylactic acid (medicinal excipients) |
|
|
 |
Triacetin (triacetate glyceride) (medicinal excipients) |
|
|
 |
Egg yolk lecithin (medicinal excipients) |
|
|
 |
Magnesium oxide (oral) (medicinal excipients) |
|
|
 |
Lanolin (medicinal excipients) |
|
|
 |
Trihydrate medicinal sodium acetate |
|
|
 |
Pharmaceutical grade poloxamer 188F68 |
|
|
 |
Medicinal hydrochloric acid |
|
|
 |
Carbomer homopolymer C type 980 NF (medicinal excipient) |
|
|
 |
85% glycerin (pharmaceutical excipient) |
|
|
 |
Docusate sodium (medicinal excipients) |
|
|
 |
Cetyl alcohol (medicinal excipients) |
|
|
 |
Sodium stearyl fumarate (medicinal excipients) |
|
|
 |
Anhydrous medicinal sodium acetate |
|
|
 |
Pharmaceutical grade polyethylene glycol 1500 |
|
|
 |
Paraffin wax (SASOLWAX 2697) (medicinal excipients) |
|
|
 |
Liquid paraffin (medicinal excipients) |
|
|
 |
Hydrogenated castor oil (medicinal excipients) |
|
|
 |
ACETIC ACID-AMMONIUM ACETATE |
|
C4H11NO4 |
 |
Glycolide lactide copolymer (7525) (medicinal excipients) |
|
|
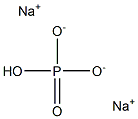 |
Disodium hydrogen phosphate (for injection) (medicinal excipients) |
|
HNa2O4P |
 |
Pharmaceutical grade povidone K90 |
|
|
 |
Glycolide lactide copolymer (5050) (medicinal excipients) |
|
|
 |
Glycolide lactide copolymer (5050) (medicinal excipients) |
|
|
 |
Sulfobutyl betacyclodextrin sodium (medicinal excipients) |
|
|
 |
Methacrylic acid copolymer type C (medicinal excipients) |
|
|
 |
Sucrose core (medicinal excipients) |
|
|
 |
Cellulose acetate succinate |
|
|
 |
Azelaic acid (medicinal excipients) |
|
|
 |
Light magnesium oxide (medicinal excipients) |
|
|
 |
Gelatin hollow capsule |
|
|
 |
Light liquid paraffin (medicinal excipients) |
|
|
 |
Dextrin (pharmaceutical excipients) |
|
|
 |
Xanthan gum (medicinal excipients) |
|
|
 |
Yamanite oleate (Span 80) (medicinal excipients) |
|
|
 |
Laurel Yamanashi (Span 20) (medicinal excipients) |
|
|
 |
Medicinal calcium sulfate |
|
|
 |
Glycolide lactide copolymer (7525) (medicinal excipients) |
|
|
 |
Carbomer homopolymer C type 980 NF (medicinal excipient) |
|
|