
1-benzyl-4-(1-methyl-1H-tetrazol-5-yl)piperidine synthesis
- Product Name:1-benzyl-4-(1-methyl-1H-tetrazol-5-yl)piperidine
- CAS Number:1401728-91-3
- Molecular formula:C14H19N5
- Molecular Weight:257.33

280111-63-9
4 suppliers
inquiry

1401728-91-3
7 suppliers
inquiry
Yield:-
Reaction Conditions:
Stage #1: 1-benzylpiperidine-4-carboxylic acid methylamidewith phosphorus pentachloride in dichloromethane at 20; for 5 h;Cooling with ice;
Stage #2: with trimethylsilylazide in dichloromethane at -5 - 20; for 3 h;
Steps:
38.2.B Step B: 1-benzyl-4-(1-methyl-1H-tetrazol-5-yl)-piperidine (37)
A solution of 1-benzyl-piperidine-4-carboxylic acid methylamide (36) (853 mg, 3.67 mmol) in dichloromethane (10 ml) was cooled in an ice-bath and phosphorous pentachloride (841 mg, 4 mmol) was added dropwise. The mixture was stirred at rt for 5 h, cooled to -5° C. and trimethylsilyl azide (387 ul, 3.67 mmol) was added dropwise. The reaction mixture was stirred for 3 h at rt and was then quenched by addition of sat. aqueous NaHCO3 solution. The organic layer was washed with water and brine, dried over Na2 SO4 and evaporated to yield a colorless solid. MS (ESI): 258 [M+H]+, 1H-NMR (DMSO-d6, 500 MHz) δ (ppm): 7.31-7.38 (m, 5H), 4.01 (s, 3H), 3.33 (br. s, 2H), 3.07 (br. m, 5H), 1.75-2.0 (br. m, 4H).
References:
US2012/252778,2012,A1 Location in patent:Paragraph 0346-0347
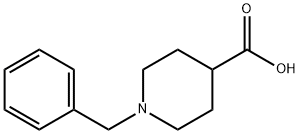
10315-07-8
133 suppliers
$10.00/250mg

1401728-91-3
7 suppliers
inquiry