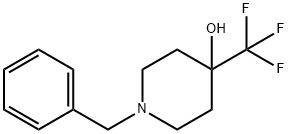
1-Benzyl-4-trifluoromethyl-piperidin-4-ol synthesis
- Product Name:1-Benzyl-4-trifluoromethyl-piperidin-4-ol
- CAS Number:373603-70-4
- Molecular formula:C13H16F3NO
- Molecular Weight:259.27
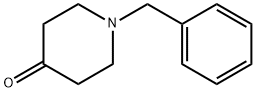
3612-20-2
460 suppliers
$6.00/10g
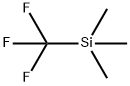
81290-20-2
421 suppliers
$13.00/1g

373603-70-4
9 suppliers
inquiry
Yield:373603-70-4 100%
Reaction Conditions:
Stage #1: 1-phenylmethyl-4-piperidone;(trifluoromethyl)trimethylsilanewith cesium fluoride in tetrahydrofuran at -5 - 20; for 2.5 h;
Stage #2: with hydrogenchloride in tetrahydrofuran;water; for 1 h;
Stage #3: with potassium carbonate in tetrahydrofuran;water;
Steps:
IP 1a.
IP 1a. 1-benzyl-4-trifluoromethylpiperidin-4-ol 0.38 g (151.90 mmol) of cesium fluoride was added to a solution of 4.63 mL (25.00 mmol) 1-benzylpiperazine-4-one in 100 mL of THF, the mixture was cooled to -5° C., 12.50 mL (142.20 mmol) trimethyltrifluoromethylsilane (2.5 M in THF) was slowly added, and the mixture was stirred for 1.5 hours at -5° C. Another 6.25 mL (71.1 mmol) of trimethyltrifluoromethylsilane was added and the mixture was stirred for 1 hour at 20° C. 100 mL of IN aqueous HCl was added and the mixture was stirred for 1 hour. The reaction mixture was made alkaline by the addition of saturated aqueous potassium carbonate solution and the phases were separated. The organic phase was dried over sodium sulfate and evaporated down in vacuo. The crude product was used in the next reaction step without further purification. Yield: 6.48 g (quant. yield); C13H16F3NO (M=259.267); calc.: molpeak (M+H)+: 260; found: molpeak (M+H)+: 260; HPLC-MS: 4.15 minutes (method A).
References:
US2005/234101,2005,A1 Location in patent:Page/Page column 10