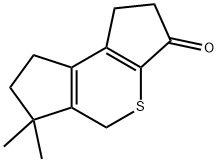
6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one synthesis
- Product Name:6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one
- CAS Number:1346672-68-1
- Molecular formula:C12H14OS
- Molecular Weight:206.3
![3-chloro-1-(5,5-dimethyl-5,6-dihydro-4H-cyclopenta[b]thiophen-2-yl)propan-1-one](/CAS/20180703/GIF/1346672-67-0.gif)
1346672-67-0
1 suppliers
inquiry
![6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one](/CAS/20180703/GIF/1346672-68-1.gif)
1346672-68-1
4 suppliers
inquiry
Yield:1346672-68-1 683 mg
Reaction Conditions:
Stage #1: 3-chloro-1-(5,5-dimethyl-5,6-dihydro-4H-cyclopenta[b]thiophen-2-yl)propan-1-onewith sulfuric acid at 90;
Stage #2: with dipotassium hydrogenphosphate in water;Cooling with acetone-dry ice;
Steps:
113f Example 113f 6,6-Dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one 113f
A 100-mL single-neck round-bottomed flask equipped with a magnetic stirrer was charged with crude 113e (2.86 g, 10.0 mmol presuming quantitative yield) and 98% sulfuric acid.
The reaction mixture was heated in a 90° C. oil bath overnight.
The reaction mixture was placed into an ice/acetone bath, and a cold (5° C.) solution of dipotassium hydrogen phosphate (105 g, 0.603 mol) in water (300 mL) was added in one portion.
The resulting mixture was shaken with ethyl acetate (300 mL) and filtered.
The filter cake was washed with ethyl acetate (100 mL).
The ethyl acetate layer of the filtrate was separated, dried over sodium sulfate and evaporated under reduced pressure on a rotary evaporator.
the resulting residue was purified by flash column chromatography (silica, 80:20 hexanes/ethyl acetate) to afford 113f in 37% yield over two steps (683 mg) as an amorphous brown solid: mp 60-62° C.; 1H NMR (500 MHz, CDCl3) δ 2.92-2.87 (m, 4H), 2.79 (s, 2H), 2.53 (s, 2H), 1.26 (s, 6H); MS (ESI+) m/z 207.0 (M+H)
References:
US2013/116262,2013,A1 Location in patent:Paragraph 0295; 0296
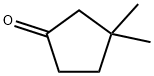
20500-49-6
59 suppliers
$27.00/250mg
![6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one](/CAS/20180703/GIF/1346672-68-1.gif)
1346672-68-1
4 suppliers
inquiry
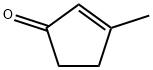
2758-18-1
244 suppliers
$9.00/1g
![6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one](/CAS/20180703/GIF/1346672-68-1.gif)
1346672-68-1
4 suppliers
inquiry
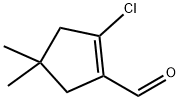
1346672-63-6
14 suppliers
inquiry
![6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one](/CAS/20180703/GIF/1346672-68-1.gif)
1346672-68-1
4 suppliers
inquiry
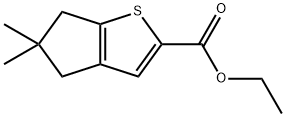
1346672-62-5
6 suppliers
inquiry
![6,6-dimethyl-1,2,6,7-tetrahydrodicyclopenta[b,d]thiophen-3(5H)-one](/CAS/20180703/GIF/1346672-68-1.gif)
1346672-68-1
4 suppliers
inquiry