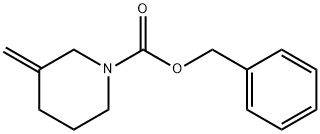
1-Piperidinecarboxylic acid, 3-Methylene-, phenylMethyl ester synthesis
- Product Name:1-Piperidinecarboxylic acid, 3-Methylene-, phenylMethyl ester
- CAS Number:138163-15-2
- Molecular formula:C14H17NO2
- Molecular Weight:231.29
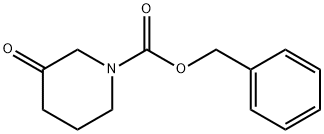
61995-20-8
158 suppliers
$29.00/5g
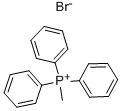
1779-49-3
580 suppliers
$5.00/10g

138163-15-2
31 suppliers
$45.00/50mg
Yield: 45%
Reaction Conditions:
Stage #1:Methyltriphenylphosphonium bromide with n-butyllithium in tetrahydrofuran at 0; for 1 h;Inert atmosphere;
Stage #2:N-(benzyloxycarbonyl)-3-piperidone in tetrahydrofuran at 0 - 20; for 1 h;Inert atmosphere;
Steps:
To a solution of PPh3CH3Br (230 g, 0.64 mol) in THF (0.8 L) is added a solution of n- BuLi (240 mL, 0.6 mol) at 0°C under N2. The mixture is stirred at 0°C for 1 h then R-7 (100 g, 0.43 mol) in THF (0.8 L) is added to the reaction mixture at 0°C. The mixture is allowed to warm to ambient temperature, stirred for 1 h, then poured into H20 and extracted with EtOAc. The organic layers are washed with brine, dried with Na2S04, concentrated and purified by flash chromatography (Si02, Hep to 25%EtOAc in Hep) to give compound R-8 (45 g, 45%). To a solution of R-8 (20.0 g, 86 mmol) in 1,4-dioxane (200 mL) is added Zn-Cu (33.2 g, 259 mmol) at rt under N2. Trichloroacetyl chloride (31.4 g, 173 mmol) in 1,4-dioxane (200 mL) is added. The mixture is allowed to warm to rt and stirred for 2 days. The mixture is treated with aqueous NaHCC>3 and extracted with EtOAc. The organic layers are washed with brine, dried with Na2S04, concentrated and purified by flash chromatography (Si02, Hep to 25 EtOAc in Hep) to give R-9 (11 g, 34%).
References:
BOEHRINGER INGELHEIM INTERNATIONAL GMBH;BENTZIEN, Joerg;BERRY, Angela;BOSANAC, Todd;BURKE, Michael, J.;DISALVO, Darren;MAO, Can;MAO, Wang;SHEN, Yue WO2015/116485, 2015, A1 Location in patent:Page/Page column 26