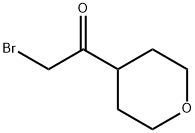
Ethanone, 2-bromo-1-(tetrahydro-2H-pyran-4-yl)- (9CI) synthesis
- Product Name:Ethanone, 2-bromo-1-(tetrahydro-2H-pyran-4-yl)- (9CI)
- CAS Number:141095-78-5
- Molecular formula:C7H11BrO2
- Molecular Weight:207.07
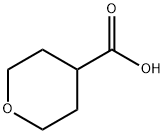
5337-03-1
296 suppliers
$10.00/1 g
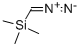
18107-18-1
210 suppliers
$33.00/5g

141095-78-5
56 suppliers
$55.00/100mg
Yield: 93.48%
Reaction Conditions:
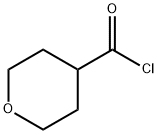
Stage #1:tetrahydro-2H-pyran-4-carboxylic acid;diazomethyl-trimethyl-silane with oxalyl dichloride in hexane;dichloromethane at -10 - 20;
Stage #2: with hydrogen bromide in hexane;dichloromethane;water at 0 - 20; for 4 h;
Steps:
5.1 Preparation 5 2-Bromo-1-tetrahydropyran-4-yl-ethanone
Method 1: (0047) Add oxalyl chloride (28.69 mL, 330.73 mmol) dropwise to a mixture of tetrahydropyran-4-carboxylic acid (39.13 g, 300.67 mmol) in DCM (250 mL) and DMF (15 drops). Stir the mixture at room temperature for 2.5 hours under nitrogen. Concentrate under reduced pressure and dissolve the residue in DCM (250 mL). Add the resulting solution dropwise to (trimethylsilyl)diazomethane (2M in hexanes, 450 mL, 900.00 mmol) at -10° C. and stir the mixture at room temperature overnight. Cool the mixture to 0° C. and add hydrobromic acid (48 wt/wt % in water, 52 mL, 462.73 mmol) dropwise. Stir the mixture at room temperature for two hours. Cool the mixture to 0° C. and add hydrobromic acid (48 wt/wt % in water, 26 mL, 231.36 mmol) dropwise. Stir the mixture at room temperature for two hours. Add water (250 mL), DCM (250 mL) and isolate the organic layer. Extract the aqueous layer with DCM (2×250 mL). Combine the organic layers and wash with saturated aqueous sodium bicarbonate solution and saturated aqueous sodium chloride. Dry over anhydrous sodium sulfate and concentrate under reduced pressure to give the title compound (58.2 g; 93.48% yield) as a brown solid. 1H NMR (300 MHz, CDCl3) δ 4.00 (m, 2H), 3.95 (s, 2H), 3.45 (m, 2H), 2.98 (m, 1H), 1.78 (m, 4H).
References:
ELi Lilly and Company;MCMILLEN, William T.;JOSEPH, Sajan;PARTHASARATHY, Saravanan;PEI, Huaxing;SAWYER, Jason Scott;BEIGHT, Douglas W.;ZHAO, Gaiying;COATES, David A.;WOLFANGEL, Craig D. US2016/96823, 2016, A1 Location in patent:Paragraph 0046-0047
137052-08-5
188 suppliers
$12.00/1g

141095-78-5
56 suppliers
$55.00/100mg
40191-32-0
128 suppliers
$18.00/1g
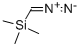
18107-18-1
210 suppliers
$33.00/5g

141095-78-5
56 suppliers
$55.00/100mg
![Ethanamine, N-[(3,5-dichlorophenyl)methylene]-2,2-diethoxy-](/CAS/20210305/GIF/1000210-73-0.gif)
1000210-73-0
0 suppliers
inquiry

141095-78-5
56 suppliers
$55.00/100mg

40191-32-0
128 suppliers
$18.00/1g

141095-78-5
56 suppliers
$55.00/100mg