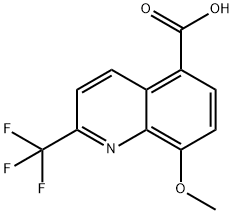
8-Methoxy-2-(trifluoroMethyl)quinoline-5-carboxylic acid synthesis
- Product Name:8-Methoxy-2-(trifluoroMethyl)quinoline-5-carboxylic acid
- CAS Number:199872-29-2
- Molecular formula:C12H8F3NO3
- Molecular Weight:271.19

848685-30-3

199872-29-2
Example 3. Preparation of 8-methoxy-2-(trifluoromethyl)quinoline-5-carboxylic acid (IX): to a solution of quinoline derivative VIII (600 g, 2.1 mol) in tetrahydrofuran (THF) (4 L) was added a solution of lithium hydroxide monohydrate (LiOH-H2O) (180 g, 4.3 mol) in water (3 L) at room temperature. The reaction mixture was stirred at room temperature for 7 hours before the THF solvent was removed by distillation under reduced pressure. Subsequently, ethyl acetate (EtOAc) (12 L) was added to the residue and then acidified by slowly adding concentrated hydrochloric acid (HCl) (330 mL). After the reaction solution was layered, the organic layer was dried with anhydrous magnesium sulfate (MgSO4). After filtration, the solvent was removed under reduced pressure to afford the target product 8-methoxy-2-(trifluoromethyl)quinoline-5-carboxylic acid (IX) (560 g, 98% yield). The structure of the product was confirmed by 1H NMR (400 MHz, DMSO-d6): δ 4.09 (s, 3H), 7.39 (d, J = 8.4 Hz, 1H), 8.11 (d, J = 9.0 Hz, 1H), 8.45 (d, J = 8.4 Hz, 1H), 9.71 (d, J = 9.0 Hz, 1H), 13.25 (br s, 1H) .

848685-30-3
1 suppliers
inquiry

199872-29-2
31 suppliers
$115.00/100mg
Yield:199872-29-2 98%
Reaction Conditions:
Stage #1: methyl 8-methoxy-2-trifluoromethyl-5-quinolinecarboxylatewith lithium hydroxide;water in tetrahydrofuran at 20; for 7 h;
Stage #2: with hydrogenchloride;water in ethyl acetate;
Steps:
3
Example 3. Preparation of the compound of formula IX :; To a mixture of the quinoline VIII (600 g, 2.1 mol) in 4 L THF at room temperature, a solution of LIOH monohydrate (180 g, 4.3 mol) in 3 L of H20 was added. After stirring at room temperature for 7 hours, the solvent THF was removed. EtOAc (12 L) was added followed by conc. HCI (330 mL). The layers were separated. The organic layer was dried over MGS04. REMOVAL of solvent gave the desired acid IX, 8-METHOXY-2-TRIFLUOROMETHYL-5- quinolinecarboxylic acid, (560 g, 98% yield). 1H NMR (400 MHz, DMSO) D 4.09 (s, 3H), 7.39 (d, J=8.4 Hz, 1H), 8. 11 (d, J=9.0 Hz, 1H), 8.45 (d, J=8.4 Hz, 1H), 9.71 (d, J=9.0 Hz, 1H), 13.25 (br, 1H)
References:
WO2005/28471,2005,A1 Location in patent:Page/Page column 22
41192-84-1
64 suppliers
$40.00/250mg

199872-29-2
31 suppliers
$115.00/100mg

41958-00-3
4 suppliers
inquiry

199872-29-2
31 suppliers
$115.00/100mg
201230-82-2
1 suppliers
inquiry

199872-02-1
2 suppliers
inquiry

199872-29-2
31 suppliers
$115.00/100mg