
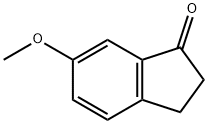
1H-INDEN-1-ONE, 6-METHOXY- synthesis
- Product Name:1H-INDEN-1-ONE, 6-METHOXY-
- CAS Number:38480-95-4
- Molecular formula:C10H8O2
- Molecular Weight:0
13623-25-1
303 suppliers
$6.00/1g

38480-95-4
5 suppliers
inquiry
Yield:38480-95-4 60%
Reaction Conditions:
Stage #1: 6-methoxy-2,3-dihydro-1H-inden-1-onewith N-Bromosuccinimide;2,2'-azobis(isobutyronitrile) in tetrachloromethane; for 2.5 h;Reflux;
Stage #2: with triethylamine in tetrachloromethane at 0;
Steps:
Representative experimental procedure for the Synthesis of benzo[c]fluorenone
General procedure: NBS (195 mg, 1.1 mmol) and AIBN (2 mg, 0.01 mmol) were added to a CCl4 solution (4 ml) of the 6-methoxyindanone (162 mg, 1 mmol). The resulting mixture was stirred at reflux temperature for 2.5 h, then cooled and filtered through Celite, which was then washed with CCl4. The filtrate was cooled to 0 °C and treated with triethylamine (0.28 mL, 2.0 mmol) overnight, then concentrated in vacuuo. Chromatography of the residue (1 : 9~1 : 4 EtOAc/hexanes) afforded 97 mg (60%) of enone 1a as the pink oil which solidified as light red solid in refrigerator. MP: 132~134°C; 1H NMR (400 MHz, CDCl3) δ ppm: 7.53 (d, J=6.0 Hz, 1H), 7.03 (d, J=2.0Hz, 1H), 6.96 (d, J=8.0 Hz, 1H), 6.77 (dd, J1=8.0 and J2=2.4 Hz, 1H), 3.81 (s, 3H); ESI-MS m/z 160.2 ([M+1]+, 100%). The 1H NMR spectral data is identical tothe literature data.[1]
References:
Zheng, Shuyan;Tan, Hongsheng;Zhang, Xiaoguang;Yu, Chunhui;Shen, Zhengwu [Tetrahedron Letters,2014,vol. 55,# 5,p. 975 - 978] Location in patent:supporting information
![1H-Indene, 5-methoxy-3-[(trimethylsilyl)oxy]-](/CAS/20210305/GIF/849405-72-7.gif)
849405-72-7
0 suppliers
inquiry

38480-95-4
5 suppliers
inquiry

537690-39-4
0 suppliers
inquiry

38480-95-4
5 suppliers
inquiry