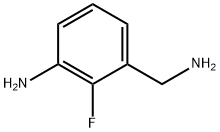
3-(aminomethyl)-2-fluoroaniline synthesis
- Product Name:3-(aminomethyl)-2-fluoroaniline
- CAS Number:946125-80-0
- Molecular formula:C7H9FN2
- Molecular Weight:140.16
![Acetamide, N-[3-(bromomethyl)-2-fluorophenyl]-](/CAS/20210305/GIF/946125-79-7.gif)
946125-79-7
0 suppliers
inquiry

946125-80-0
6 suppliers
inquiry
Yield:946125-80-0 48%
Reaction Conditions:
Stage #1: N-(3-bromomethyl-2-fluorophenyl)acetamidewith hexamethylenetetramine in chloroform; for 1.66667 h;Reflux;
Stage #2: with hydrogenchloride in water at 20; for 30 h;
Steps:
8 3-Aminoethyl-2-fluoro-phenylamine
A solution of hexamethyltetramine (595 mg,4.24 mmol) in chloroform (4.0 mL) was heated under reflux, and a solution of N-(3-bromomethyl-2-fluorophenyl)acetamide (950 mg, 3.86 mmol) in chloroform(8.0 mL) was then added thereto over a period of 40 minutes. The reaction mixture was further heated under reflux for 1 hour, and after returning to room temperature, the white precipitate was filtered out and washed with chloroform.Methanol (24 mL) and concentrated hydrochloric acid (3.0 mL) were added to the white precipitate, and the mixture was stirred at room temperature for 30 hours. After cooling to 0° C., the reaction mixture was rendered alkaline witha 6N sodium hydroxide aqueous solution (16.5 mL). It was then extracted with dichloromethane, and the organic extract was washed with saturated saline anddried over magnesium sulfate, and again washed with saturated saline. After drying over magnesium sulfate, the dried extract was concentrated under reducedpressure to yield a crude product, which was then purified by column chromatography to yield the title compound (337 mg, 48%) as a brown solid.
References:
KR101511396,2015,B1 Location in patent:Paragraph 3505-3508