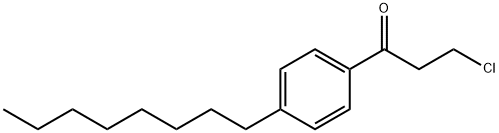
3-chloro-1-(4-octylphenyl)-preopanone synthesis
- Product Name:3-chloro-1-(4-octylphenyl)-preopanone
- CAS Number:928165-59-7
- Molecular formula:C17H25ClO
- Molecular Weight:280.83
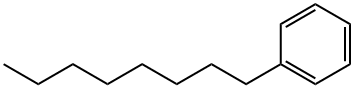
2189-60-8
219 suppliers
$30.00/20mg
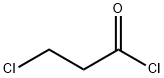
625-36-5
394 suppliers
$45.00/5g

928165-59-7
51 suppliers
$268.00/1g
Yield:928165-59-7 72%
Reaction Conditions:
with aluminum (III) chloride in dichloromethane at -10 - -5; for 3 - 5 h;Large scale;Friedel-Crafts Acylation;
Steps:
Synthesis of 3-Nitro-(4-octyl-phenyl)-propane-1-one (2)
In a reactor, octanophenone was added (5.0 kg, 24.5 mol) to CH2Cl2 (25 L). The mixture was cooled at -10 to -5 °C and was added to triethylsilane, followed by TiCl4 (4.9 kg, 25.7 mol) in CH2Cl2 (25 L) at -10 to 5 °C. The temperature was raised from 0 to 5 °C and stirred for 3-5 h. Progress of the reaction was monitored by TLC, after completion of the reaction, the mixture was filtered through hyflow bed and washed withCH2Cl2 (10.0 L). The combined organic layer was transferred to another reactor and cooled to -10 °C from -5 °C. To this was added AlCl3 (4.25 kg, 1.3 equiv.) followed by 3-chloropropionyl chloride (3.5 kg, 2.7mol) in CH2Cl2 (10.0 L) and stirred for 3-5 h at -10 to -5 °C. Reaction progress was monitored by HPLC. After completion of reaction, ice cold water was slowly added (50.0 L) at below 15 °C and stirred for 1-2 h. The organic layer was separated, washed with 5% NaHCO3 solution, brine solution and distilled below 40 °C under vacuum to get crude compound 6. To a reactor the crude compound 6, NaNO2 (2.7 kg, 39.2 mol) in DMF (30.0 L) was added and stirred at 25-35 °C for 4-6 h. Reaction progress was monitored by TLC. After completion of the reaction, H2O (50.0 L) and EtOAc (50.0 L) were added and the mixture was stirred for 15-30 min at 25-35 °C. The organic layer was separated and the aqueous layer was washed with EtOAc (10.0L). The combined organic layer was washed with brine (2 x 50.0 L) and distilled below 50 °C under vacuum to get the crude compound. To the crude compound was added n-hexane (20.0 L), cooled to -15 to -10 °C,and stirred for 45-60 min. The solid was filtered and washed with chilled n-hexane (5.0 L). Then wet cake followed by MeOH (30.0 L) was added into another reactor at 25-35 C. Then the mixture was cooled to -15 to -10 °C, stirred for 60 min, filtered and washed with chilled MeOH (5.0 L) to get the compound 2. The solid was dried at 35-40 °C under vacuum (5.06 kg, Yield: 71%).
References:
Vinigari, Krishna;Jonnada, Krishna;Mohammed, Noorjahan;Kotu, Girija Mangatayaru [Synthetic Communications,2019,vol. 49,# 1,p. 39 - 48]
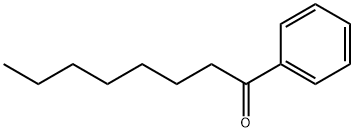
1674-37-9
131 suppliers
$36.10/5g

625-36-5
394 suppliers
$45.00/5g

928165-59-7
51 suppliers
$268.00/1g

1674-37-9
131 suppliers
$36.10/5g

928165-59-7
51 suppliers
$268.00/1g