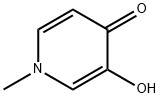
3-hydroxy-1-methyl-4(1H)-Pyridinone synthesis
- Product Name:3-hydroxy-1-methyl-4(1H)-Pyridinone
- CAS Number:50700-61-3
- Molecular formula:C6H7NO2
- Molecular Weight:125.13
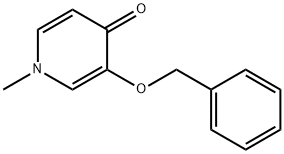
1064077-34-4

50700-61-3
The general procedure for the synthesis of 3-hydroxy-1-methyl-1H-pyridin-4-one from 1-methyl-3-benzyloxy-4(1H)-pyridinone was as follows: a mixture of 3-benzyloxy-1-methyl-1H-pyridin-4-one (14.1 g, 65.5 mmol) with 10% Pd/C (1.20 g) in methanol was placed in a Parr reactor at 50 psi The hydrogenation reaction was carried out under hydrogen pressure for 70 min. Upon completion of the reaction, the mixture was filtered through a Celite pad and the filtrate was concentrated to dryness. The residue was ground with acetone and the solid product was subsequently collected by filtration. After drying in a vacuum oven, 3-hydroxy-1-methyl-1H-pyridin-4-one was obtained as a light brown powder (7.13 g, 87% yield). The product was characterized by 1H NMR (400 MHz, DMSO-d6): δ 7.49 (br.s, 1H), 7.38 (s, 1H), 6.12 (br.s, 1H), 3.63 (s, 3H); MS-ESI (m/z): 125.7 [M + 1]+.

1064077-34-4
17 suppliers
inquiry

50700-61-3
31 suppliers
inquiry
Yield:50700-61-3 87%
Reaction Conditions:
with hydrogen;palladium 10% on activated carbon in methanol; under 2585.81 Torr; for 1.16667 h;
Steps:
22.B
A mixture of 3-benzyloxy-1-methyl-1H-pyridin-4-one (14.1 g, 65.5 mmol) and 10% Pd/C (1.20 g) in methanol was subjected to hydrogenation in a Parr apparatus under 50 psi pressure of hydrogen for 70 min. The mixture was filtered through a pad of CELITE, the filtrate was concentrated to dryness, and the residue was triturated with acetone. The solid was then collected by suction filtration. The title compound was obtained as a light-brown powder (7.13 g, 87% yield) after vacuum oven drying. 1H NMR (400 MHz, DMSO-D6) δ (ppm): 7.49 (br. s, 1H), 7.38 (s, 1H), 6.12 (br. s, 1H) and 3.63 (s, 3H); MS-ESI (m/z): 125.7 [M+1]+.
References:
US2008/242706,2008,A1 Location in patent:Page/Page column 28