
5-Chloro-1,2,3,6-tetrahydro-2,6-dioxo-4-pyrimidinecarboxylic acid synthesis
- Product Name:5-Chloro-1,2,3,6-tetrahydro-2,6-dioxo-4-pyrimidinecarboxylic acid
- CAS Number:36313-98-1
- Molecular formula:C5H3ClN2O4
- Molecular Weight:190.54
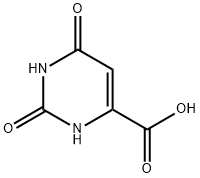
65-86-1
449 suppliers
$5.00/100mg

36313-98-1
22 suppliers
inquiry
Yield:36313-98-1 92%
Reaction Conditions:
Stage #1: orotic acidwith iron(III) chloride;acetic anhydride;acetic acid at 90 - 95;
Stage #2: with sulfuryl dichlorideReflux;Reagent/catalyst;Temperature;
Steps:
a.1 Example 1
78 g (0.50 mol) of orotic acid (B0) is added to 700 ml of acetic acid and 25 ml of acetic anhydride and a catalytic amount of ferric chloride are added, plusHeat to 90-95°C and add 108g (0.80mol) of sulfonyl chloride (FW135) dropwise. After the addition is complete, heat to reflux overnight until the methyl orotate is completely chlorinated (TLC detection) to stop the reaction. Cool the reaction solution to 10°C.The solid was collected by filtration, washed sequentially with acetic acid and water, and dried with hot air to give a yellow solid. Melting point: mp: 288° C., yielding 88.0 g (0.46 mol) of α-chloroorotic acid (B1), molar yield 92%.
References:
CN107118164,2017,A Location in patent:Page/Page column 7